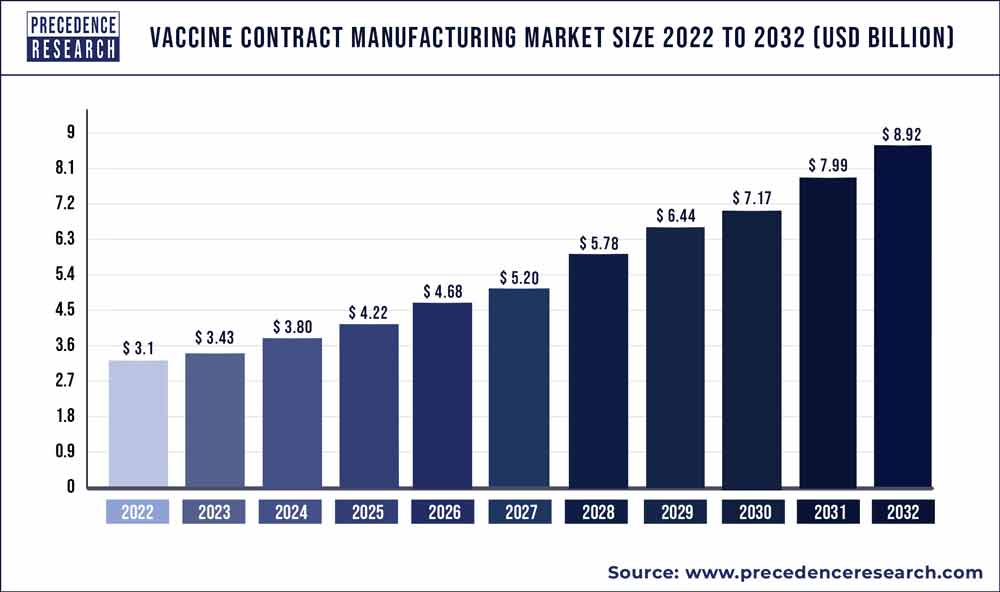
The global vaccine contract manufacturing market size was estimated to be around US$ 3.1 billion in 2022. It is projected to reach US$ 8.92 billion by 2032, indicating a CAGR of 11.2% from 2023 to 2032.
Key Takeaways
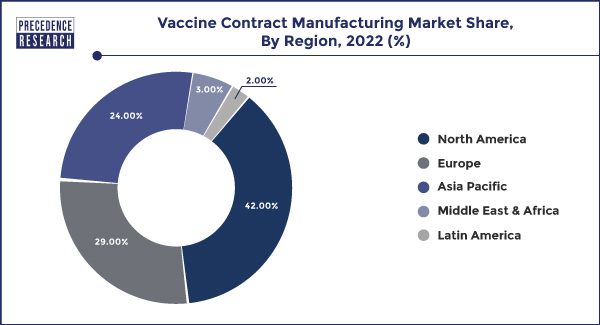
- North America led the market with the highest market share of 42% in 2022.
- Asia-Pacific is expected to grow at the fastest CAGR during the forecast period.
- By Vaccine Type, the attenuated vaccines segment has held the largest revenue share of 28% in 2022.
- By Vaccine Type, the inactivated inoculations segment is projected to expand at a remarkable CAGR of 12.7% during the projected period.
- By Workflow, the downstream segment generated more than 68% of revenue share in 2022.
- By Workflow, the upstream segment is anticipated to grow at the fastest CAGR over the projected period.
- By Application, the human-use segment held the major revenue share of 69% in 2022.
- By Application, the veterinary segment is estimated to grow at a noteworthy CAGR of 13.3% over the predicted period.
Vaccine Contract Manufacturing Marke Overview:
The Vaccine Contract Manufacturing Market is a dynamic sector within the pharmaceutical industry that involves outsourcing the production of vaccines to third-party manufacturers. This practice allows pharmaceutical companies and organizations to leverage external expertise and facilities to meet the growing demand for vaccines globally. The market encompasses a wide range of services, including development, manufacturing, packaging, and distribution of vaccines. As the need for vaccination continues to rise, fueled by emerging infectious diseases and the ongoing efforts to combat existing ones, the demand for contract manufacturing services in the vaccine industry has witnessed significant growth.
Get a Sample: https://www.precedenceresearch.com/sample/3421
Vaccine Contract Manufacturing Market Scope
| Report Coverage | Details |
| Growth Rate from 2023 to 2032 | CAGR of 11.2% |
| Market Size in 2023 | USD 3.43 Billion |
| Market Size by 2032 | USD 8.92 Billion |
| Largest Market | North America |
| Base Year | 2022 |
| Forecast Period | 2023 to 2032 |
| Segments Covered | By Vaccine Type, By Workflow, and By Application |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Read More: Tuberculosis Diagnostics Market Size To Increase USD 3.67 Billion By 2032
Growth Factors:
Several factors contribute to the growth of the Vaccine Contract Manufacturing Market. Firstly, the increasing prevalence of infectious diseases and the need for timely vaccine development have led pharmaceutical companies to seek efficient and scalable manufacturing solutions. Outsourcing to contract manufacturers enables these companies to accelerate the production process, reduce costs, and navigate regulatory challenges effectively. Additionally, the complexity of vaccine production, particularly for novel and advanced vaccine technologies, has driven the industry towards specialized contract manufacturing organizations with the necessary expertise. The global push for vaccination, exemplified by initiatives such as the COVAX program, further propels the demand for contract manufacturing services to ensure widespread and equitable vaccine distribution.
Furthermore, the flexibility offered by contract manufacturing allows companies to adapt quickly to changes in vaccine demand and emerging health threats. The strategic alliances formed between pharmaceutical companies and contract manufacturers contribute to a more resilient and responsive vaccine supply chain. As technology continues to advance, the incorporation of innovative manufacturing processes and techniques by contract manufacturers enhances the overall efficiency and scalability of vaccine production.
Region Snapshot:
The Vaccine Contract Manufacturing Market exhibits a global presence, with key regions playing vital roles in its growth. North America, particularly the United States and Canada, holds a significant share of the market due to the presence of major pharmaceutical companies and advanced manufacturing infrastructure. Europe is another prominent region, with established contract manufacturing organizations and a focus on research and development in vaccine technologies.
The Asia-Pacific region is emerging as a key player, driven by the increasing demand for vaccines, a growing pharmaceutical industry, and a skilled workforce. As the need for vaccines extends to developing nations, regions in Latin America, Africa, and the Middle East are becoming important markets for vaccine contract manufacturing, creating opportunities for both local and international players. The interplay of these regional dynamics contributes to the overall expansion and diversification of the Vaccine Contract Manufacturing Market on a global scale.
Reasons to Purchase this Report:
- Comprehensive market segmentation analysis incorporating qualitative and quantitative research, considering the impact of economic and policy factors.
- In-depth regional and country-level analysis, examining the demand and supply dynamics that influence market growth.
- Market size in USD million and volume in million units provided for each segment and sub-segment.
- Detailed competitive landscape, including market share of major players, recent projects, and strategies implemented over the past five years.
- Comprehensive company profiles encompassing product offerings, key financial information, recent developments, SWOT analysis, and employed strategies by major market players.
Recent Developments
- In July 2023, Merck unveiled plans to expand its laboratory space and production capabilities in Lenexa, Kansas, USA. This expansion aimed to enhance its capacity for manufacturing cell culture media and accelerate the production of innovative dry powder media, crucial for processes like vaccine manufacturing and gene therapy.
- FUJIFILM Corporation, in June 2023, announced the establishment of a commercial office in Tokyo. This office was strategically positioned to offer improved sales and customer support services, particularly catering to Asia-based pharmaceutical and biotechnology firms, for their contract development and manufacturing needs in biologics and advanced therapies.
- Lonza made a significant move in June 2023 by acquiring Synaffix B.V. This acquisition was focused on bolstering Lonza’s bioconjugate capabilities through integration with Synaffix’s technology, further strengthening their Center of Excellence dedicated to bioconjugate technology development.
- Lonza and Vertex Pharmaceuticals Incorporated, also in June 2023, entered into a strategic partnership to scale up manufacturing and support the commercial production of Vertex’s T1D cell therapy portfolio. They additionally planned to jointly invest in the construction of a dedicated facility in Portsmouth, New Hampshire, USA.
- In April 2023, FUJIFILM Diosynth Biotechnologies announced its acquisition of 41 acres of adjacent land to its existing RTP campus in North Carolina. This expansion aimed to accelerate their production capabilities for biologics manufacturing and development.
- Cytovance Biologics, Inc. partnered with Phenotypeca Limited in March 2023 to provide optimized strains of Saccharomyces cerevisiae for biopharmaceutical manufacturing. This collaboration enhanced Cytovance Biologics’ leadership in the production of microbial-derived active pharmaceutical ingredients (API) and expanded Phenotypeca’s market presence.
- IDT Biologika joined forces with CanVirex, a biotechnology company, to manufacture oncolytic measles virus-based therapeutics for anti-cancer treatment under cGMP conditions. IDT utilized its advanced manufacturing infrastructure for this collaboration.
- In January 2023, Ajinomoto Co., Inc. entered into a partnership with Exelixis, Inc. to jointly discover and develop novel Antibody-Drug Conjugates (ADCs) for cancer treatment. The collaboration leveraged Ajinomoto’s AJICAP technology to create advanced ADCs with improved efficacy and reduced toxicity.
Vaccine Contract Manufacturing Market Players
- Lonza
- FUJIFILM Diosynth Biotechnologies U.S.A., Inc.
- Ajinomoto Althea, Inc.
- Merck KgaA
- Cytovance Biologics
- Catalent, Inc.
- IDT Biologika GmbH
- Albany Molecular Research, Inc.
- PRA Health Sciences
- ICON plc.
- Pharmaceutical Product Development, LLC
- Cobra Bio
- Paragon Bioservices, Inc.
Market Segmentations
By Vaccine Type
- Attenuated
- Inactivated
- Subunit-based
- Toxoid-based
- DNA-based
By Workflow
- Downstream
- Upstream
By Application
- Human Use
- Veterinary
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
TABLE OF CONTENT
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology (Premium Insights)
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Vaccine Contract Manufacturing Market
5.1. COVID-19 Landscape: Vaccine Contract Manufacturing Industry Impact
5.2. COVID 19 – Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Vaccine Contract Manufacturing Market, By Vaccine Type
8.1. Vaccine Contract Manufacturing Market, by Vaccine Type, 2023-2032
8.1.1 Attenuated
8.1.1.1. Market Revenue and Forecast (2020-2032)
8.1.2. Inactivated
8.1.2.1. Market Revenue and Forecast (2020-2032)
8.1.3. Subunit-based
8.1.3.1. Market Revenue and Forecast (2020-2032)
8.1.4. Toxoid-based
8.1.4.1. Market Revenue and Forecast (2020-2032)
8.1.5. DNA-based
8.1.5.1. Market Revenue and Forecast (2020-2032)
Chapter 9. Global Vaccine Contract Manufacturing Market, By Workflow
9.1. Vaccine Contract Manufacturing Market, by Workflow, 2023-2032
9.1.1. Downstream
9.1.1.1. Market Revenue and Forecast (2020-2032)
9.1.2. Upstream
9.1.2.1. Market Revenue and Forecast (2020-2032)
9.1.3. Electrolyte
9.1.3.1. Market Revenue and Forecast (2020-2032)
9.1.4. Separator
9.1.4.1. Market Revenue and Forecast (2020-2032)
9.1.5. Others
9.1.5.1. Market Revenue and Forecast (2020-2032)
Chapter 10. Global Vaccine Contract Manufacturing Market, By Application
10.1. Vaccine Contract Manufacturing Market, by Application, 2023-2032
10.1.1. Human Use
10.1.1.1. Market Revenue and Forecast (2020-2032)
10.1.2. Veterinary
10.1.2.1. Market Revenue and Forecast (2020-2032)
Chapter 11. Global Vaccine Contract Manufacturing Market, Regional Estimates and Trend Forecast
11.1. North America
11.1.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.1.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.1.3. Market Revenue and Forecast, by Application (2020-2032)
11.1.4. U.S.
11.1.4.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.1.4.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.1.4.3. Market Revenue and Forecast, by Application (2020-2032)
11.1.5. Rest of North America
11.1.5.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.1.5.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.1.5.3. Market Revenue and Forecast, by Application (2020-2032)
11.2. Europe
11.2.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.2.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.2.3. Market Revenue and Forecast, by Application (2020-2032)
11.2.4. UK
11.2.4.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.2.4.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.2.4.3. Market Revenue and Forecast, by Application (2020-2032)
11.2.5. Germany
11.2.5.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.2.5.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.2.5.3. Market Revenue and Forecast, by Application (2020-2032)
11.2.6. France
11.2.6.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.2.6.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.2.6.3. Market Revenue and Forecast, by Application (2020-2032)
11.2.7. Rest of Europe
11.2.7.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.2.7.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.2.7.3. Market Revenue and Forecast, by Application (2020-2032)
11.3. APAC
11.3.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.3.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.3.3. Market Revenue and Forecast, by Application (2020-2032)
11.3.4. India
11.3.4.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.3.4.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.3.4.3. Market Revenue and Forecast, by Application (2020-2032)
11.3.5. China
11.3.5.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.3.5.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.3.5.3. Market Revenue and Forecast, by Application (2020-2032)
11.3.6. Japan
11.3.6.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.3.6.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.3.6.3. Market Revenue and Forecast, by Application (2020-2032)
11.3.7. Rest of APAC
11.3.7.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.3.7.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.3.7.3. Market Revenue and Forecast, by Application (2020-2032)
11.4. MEA
11.4.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.4.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.4.3. Market Revenue and Forecast, by Application (2020-2032)
11.4.4. GCC
11.4.4.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.4.4.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.4.4.3. Market Revenue and Forecast, by Application (2020-2032)
11.4.5. North Africa
11.4.5.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.4.5.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.4.5.3. Market Revenue and Forecast, by Application (2020-2032)
11.4.6. South Africa
11.4.6.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.4.6.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.4.6.3. Market Revenue and Forecast, by Application (2020-2032)
11.4.7. Rest of MEA
11.4.7.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.4.7.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.4.7.3. Market Revenue and Forecast, by Application (2020-2032)
11.5. Latin America
11.5.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.5.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.5.3. Market Revenue and Forecast, by Application (2020-2032)
11.5.4. Brazil
11.5.4.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.5.4.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.5.4.3. Market Revenue and Forecast, by Application (2020-2032)
11.5.5. Rest of LATAM
11.5.5.1. Market Revenue and Forecast, by Vaccine Type (2020-2032)
11.5.5.2. Market Revenue and Forecast, by Workflow (2020-2032)
11.5.5.3. Market Revenue and Forecast, by Application (2020-2032)
Chapter 12. Company Profiles
12.1. Lonza
12.1.1. Company Overview
12.1.2. Product Offerings
12.1.3. Financial Performance
12.1.4. Recent Initiatives
12.2. FUJIFILM Diosynth Biotechnologies U.S.A., Inc.
12.2.1. Company Overview
12.2.2. Product Offerings
12.2.3. Financial Performance
12.2.4. Recent Initiatives
12.3. Ajinomoto Althea, Inc.
12.3.1. Company Overview
12.3.2. Product Offerings
12.3.3. Financial Performance
12.3.4. Recent Initiatives
12.4. Merck KgaA
12.4.1. Company Overview
12.4.2. Product Offerings
12.4.3. Financial Performance
12.4.4. Recent Initiatives
12.5. Cytovance Biologics
12.5.1. Company Overview
12.5.2. Product Offerings
12.5.3. Financial Performance
12.5.4. Recent Initiatives
12.6. Catalent, Inc.
12.6.1. Company Overview
12.6.2. Product Offerings
12.6.3. Financial Performance
12.6.4. Recent Initiatives
12.7. IDT Biologika GmbH
12.7.1. Company Overview
12.7.2. Product Offerings
12.7.3. Financial Performance
12.7.4. Recent Initiatives
12.8. Albany Molecular Research, Inc.
12.8.1. Company Overview
12.8.2. Product Offerings
12.8.3. Financial Performance
12.8.4. Recent Initiatives
12.9. PRA Health Sciences
12.9.1. Company Overview
12.9.2. Product Offerings
12.9.3. Financial Performance
12.9.4. Recent Initiatives
12.10. ICON plc.
12.10.1. Company Overview
12.10.2. Product Offerings
12.10.3. Financial Performance
12.10.4. Recent Initiatives
Chapter 13. Research Methodology
13.1. Primary Research
13.2. Secondary Research
13.3. Assumptions
Chapter 14. Appendix
14.1. About Us
14.2. Glossary of Terms
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com