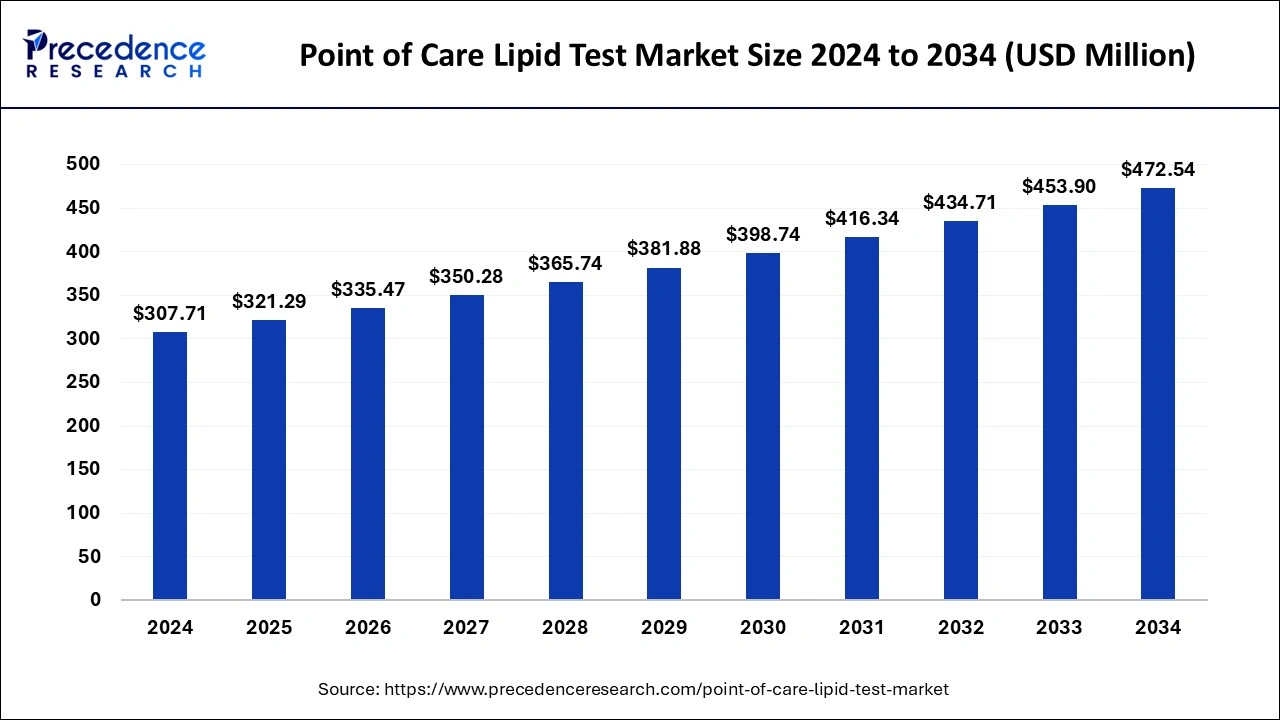
The global point of care lipid test market size was estimated at USD 294.70 million in 2023 and is projected to attain around USD 453.90 million by 2033, growing at a CAGR of 4.41% from 2024 to 2033.
Key Points
- North America held a significant share of in the point of care lipid test market in 2023 with 38.6%.
- Asia Pacific is anticipated to experience the highest growth in the upcoming years.
- By product, the consumables type segment has accounted market share of 57% in 2023.
- By application, in 2023, the endogenous hyperlipidemia segment dominated the market with 46% revenue share.
- By end use, the diagnostic centers segment has held largest market share of 56% in 2023.
The point-of-care lipid test market encompasses the production and distribution of diagnostic devices designed to measure lipid levels in patients’ blood quickly and accurately at the point of care. Lipids, which include cholesterol and triglycerides, play a significant role in various health conditions, including cardiovascular diseases. Regular lipid testing helps monitor these levels and can aid in the prevention and management of conditions such as atherosclerosis and heart attacks.
The market for point-of-care lipid tests is growing due to the increasing prevalence of cardiovascular diseases and the growing need for early diagnosis and monitoring. The convenience and speed of these tests, which can be performed in non-laboratory settings such as clinics and even patients’ homes, have contributed to their popularity. These tests provide immediate results, allowing healthcare providers to make quick decisions regarding a patient’s treatment plan.
Get a Sample: https://www.precedenceresearch.com/sample/4076
Growth Factors
- Rising Prevalence of Cardiovascular Diseases: As cardiovascular diseases become more common worldwide, the demand for lipid testing has increased. Point-of-care lipid tests offer a convenient and rapid way to monitor patients’ health.
- Technological Advancements: Improvements in point-of-care testing technology have led to more accurate and efficient tests, driving market growth. These advancements include enhanced test sensitivity and ease of use.
- Increasing Awareness of Preventive Healthcare: More people are taking a proactive approach to their health, including monitoring their lipid levels to prevent potential issues. This has led to greater demand for point-of-care lipid tests.
- Regulatory Support and Reimbursement Policies: Governments and healthcare organizations are supporting the use of point-of-care testing through favorable reimbursement policies and regulations, encouraging their adoption.
- Aging Population: An aging global population is more likely to suffer from cardiovascular diseases, which increases the need for lipid monitoring and testing.
Region Insights
- North America: This region is the largest market for point-of-care lipid tests due to its advanced healthcare infrastructure, higher healthcare expenditure, and awareness of preventive health measures. The presence of key market players also contributes to the region’s growth.
- Europe: Europe has a strong healthcare system and a high prevalence of cardiovascular diseases. The market in this region benefits from robust regulatory support and public awareness campaigns on cardiovascular health.
- Asia-Pacific: The Asia-Pacific region is experiencing significant growth in the point-of-care lipid test market due to increasing healthcare awareness and a rising prevalence of cardiovascular diseases. Countries like China and India are investing heavily in healthcare infrastructure, further boosting market growth.
- Latin America and Middle East & Africa: These regions are gradually adopting point-of-care lipid tests due to improving healthcare systems and an increasing focus on preventive healthcare. However, market penetration remains relatively low compared to other regions.
Point of Care Lipid Test Market Scope
| Report Coverage | Details |
| Global Market Size in 2023 | USD 294.70 Million |
| Global Market Size in 2024 | USD 307.71 Million |
| Global Market Size by 2033 | USD 453.90 Million |
| Growth Rate from 2024 to 2033 | CAGR of 4.41% |
| Largest Market | North America |
| Base Year | 2023 |
| Forecast Period | 2024 to 2033 |
| Segments Covered | By Product Type, By Application, and By End-user |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Drivers
- Ease of Use and Accessibility: Point-of-care lipid tests are user-friendly and can be performed by healthcare providers with minimal training, allowing for widespread use in various healthcare settings.
- Quick Results: These tests provide results within minutes, enabling healthcare providers to make immediate decisions about a patient’s treatment plan.
- Home Testing: The rise of home-based healthcare has increased the demand for point-of-care lipid tests that patients can use independently at home.
- Reducing Healthcare Costs: Point-of-care lipid tests can help reduce overall healthcare costs by allowing for earlier diagnosis and monitoring of patients, potentially preventing costly complications.
- Customization and Portability: Many point-of-care lipid test devices are portable and customizable, enabling healthcare providers to tailor testing to individual patient needs.
Opportunities
- Emerging Markets: Developing regions present opportunities for market expansion as healthcare infrastructure improves and awareness of cardiovascular health grows.
- Product Innovation: Continuous improvements in technology open opportunities for new and innovative point-of-care lipid testing devices with enhanced capabilities.
- Telemedicine Integration: The integration of point-of-care lipid tests with telemedicine platforms can improve remote monitoring and consultation, further driving demand.
- Partnerships and Collaborations: Collaborations between manufacturers, research institutions, and healthcare providers can lead to new product development and market expansion.
- Regulatory Approvals: Gaining approvals from regulatory agencies in emerging markets can provide a competitive edge and open up new revenue streams.
Challenges
- Cost of Devices: The initial cost of point-of-care lipid test devices can be high, potentially limiting access in some regions.
- Regulatory Hurdles: Stringent regulatory requirements for diagnostic devices can slow down product development and market entry.
- Quality Control and Accuracy: Ensuring the accuracy and reliability of point-of-care lipid tests is critical for patient safety and regulatory approval.
- Competition from Laboratory Testing: Traditional laboratory-based lipid testing may still be preferred in some settings due to established infrastructure and familiarity.
- Limited Awareness: In certain regions, limited awareness of the benefits of point-of-care lipid testing may hinder market growth.
The global point of care lipid test market size was valued at USD 294.70 million in 2023 and is anticipated to reach around USD 453.90 million by 2033, growing at a CAGR of 4.41% from 2024 to 2033. The rising prevalence of target diseases such as cardiovascular diseases, diabetes, and dyslipidemia is identified as a key driving factor of the point of care lipid test market.
Read Also: Electric Vehicle Thermal Management Market Size, Share, Report By 2033
Recent Developments
- In October 2022 Genes2Me Pvt. Ltd launched Rapi-Q- Point of Care RT PCR solution for human papillomavirus (HPV) and tuberculosis. The device is easy to use and gives faster results in less than 45 minutes. This CE-IVD-marked POC solution delivers superior performance, high sensitivity, and stable detection.
- In March 2022, Visby Medical announced that it received funding of USD 25.5 million from the U.S. Biomedical Advanced Research and Development Authority to develop a rapid flu-COVID-19 PCR test for home use. At present, the test is in the under-developing phase, and the design is ready as a PCR device that can detect COVID-19, influenza A, and B from a single sample.
- In March 2022, Canada-based Company BioLytical Laboratories Inc. received a CE marking for the iStatis COVID-19 Antigen Home Test.
- In May 2022, Qiagen Inc. launched the NeuMoDxHSV 1/2 Quant Assay for the quantification and differentiation of herpes simplex virus type 1 (HSV-1) DNA and herpes simplex virus type 2 (HSV-2) with approval from the European Commission. The emergence of this assay is allowing the company to expand its product portfolio in laboratory testing, which ultimately helps the market grow owing to the innovative tests.
- In March 2022, Mindray launched the BC-700 Series, a hematology analyzer series that assists in both blood count and erythrocyte sedimentation rate tests.
Point of Care Lipid Test Market Com[panies
- Callegari Sinocare Inc.
- Abbott Laboratories
- Mico Bio Med
- Nova Biomedical Corporation
- VivaChek Biotech (Hangzhou) Co., Ltd.
- F. Hoffmann-La Roche Ltd.
- Zoetis Inc.
- Menarini Group
- SD Biosensor, Inc.
Segments Covered in the Report
By Product Type
- Devices
- Consumables
By Application
- Endogenous Hyperlipemia
- Combined Hyperlipidemia
- Familial Hypercholesterolemia
- Others
By End-user
- Hospitals And Clinics
- Diagnostic Laboratories
- Others
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com
Blog: https://www.expresswebwire.com/
Blog: https://www.uswebwire.com/
Blog: https://www.dailytechbulletin.com/
Blog: https://www.autoindustrybulletin.com/