Drivers
- Affordability of Generics – Lower costs compared to branded drugs make them accessible to a wider population.
- Patent Expirations – Numerous blockbuster drug patents are expiring, opening the door for generic alternatives.
- Government Initiatives – Policies promoting generic drug usage to reduce healthcare expenditure.
- Rising Chronic Disease Prevalence – Increasing cases of cancer, diabetes, and cardiovascular diseases are boosting demand.
Restraints
- Regulatory Hurdles – Stringent approval processes by agencies such as the FDA and EMA can delay product launches.
- Patent Protection – Ongoing exclusivity periods for branded drugs limit immediate generic entry.
Opportunities
- Emerging Markets – Countries like India and China offer high-growth potential due to large patient pools and supportive policies.
- Digital Pharmacies & Supply Chain Automation – Enhanced efficiency, improved patient outreach, and real-time demand forecasting.
Regional Insights
North America
North America accounted for the largest revenue share in 2024, with the U.S. market valued at USD 139.03 billion, projected to reach USD 231.69 billion by 2034 at a CAGR of 5.24%. Growth is driven by the strong presence of pharmaceutical companies, high regulatory standards, and the affordability of generics. The U.S. also benefits from global partnerships, accounting for roughly 30% of India’s total pharma exports.
Asia-Pacific
The Asia-Pacific region is poised for the fastest growth due to rising healthcare demand, supportive government initiatives, and the dominance of India and China in low-cost generic production. India remains a global leader, producing about 20% of global generic drug volume and meeting 40% of
U.S. generic demand
Europe, Latin America, and MEA
Europe’s demand is supported by universal healthcare systems adopting generics to control costs. Latin America and the Middle East & Africa (MEA) are experiencing rising generic drug uptake due to increasing chronic disease prevalence and improved healthcare infrastructure.
Generic Drugs Market Growth Factors
-
Low Cost Compared to Branded Drugs
Generic drugs offer a far more affordable alternative to branded medications, making them an essential solution for patients in both developed and developing nations. Their cost-effectiveness helps reduce overall healthcare expenditure while maintaining treatment efficacy. -
Patent Expirations of Branded Drugs
The expiration of patents on several high-revenue branded drugs creates significant opportunities for generic manufacturers to launch bioequivalent alternatives. This factor has been a major driver in expanding generic drug portfolios globally. -
Government and Regulatory Initiatives
Governments and regulatory bodies worldwide are actively promoting the adoption of generics through healthcare reforms, policy incentives, and bulk procurement programs aimed at improving accessibility and lowering costs.
Market Scope
| Report Highlights | Details |
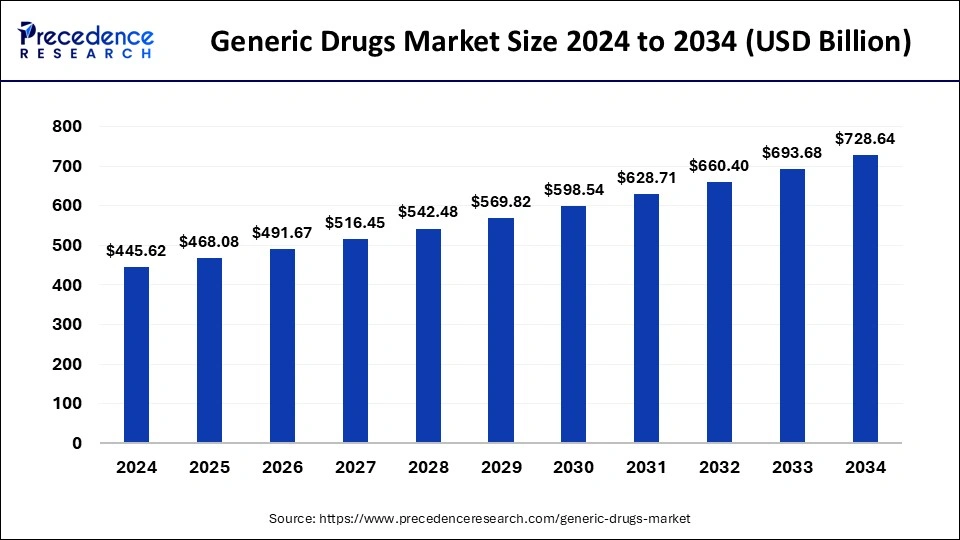
| Market Size in 2024 | USD 445.62 billion |
| Market Size in 2025 | USD 468.08 Billion |
| Market Size by 2034 | USD 728.64 Billion |
| Growth Rate from 2025 to 2034 | CAGR of 5.04% |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Drug, Brand, Route of Drug Administration, Therapeutic Application, Distribution Channel |
| Regional Scope | North America, Europe, Asia Pacific, Latin America, Middle East & Africa (MEA) |
Segment Overview
- By Brand – Pure generics led with over half the market share in 2024; branded generics remain important for patient recognition and loyalty.
- By Route of Administration – Oral generics dominate due to ease of use, while injectables are the fastest-growing segment for specialized treatments.
- By Drug Type – Simple generics currently lead, but super generics are expected to grow rapidly due to improved formulations.
- By Therapeutic Application – Oncology is the fastest-growing application, driven by the high cost of branded cancer treatments.
- By Distribution Channel – Retail pharmacies dominate, but hospital pharmacies are expected to post robust growth.
Recent Industry Developments
- June 2025 – Advent International invested USD 175 million in Felix Pharmaceuticals, expanding the generic veterinary medicines segment.
- August 2025 – Zenara Pharma received FDA approval for Sertraline Hydrochloride capsules as the first generic equivalent to Almatica Pharma’s product, with Competitive Generic Therapy designation.
- August 2025 – Sandoz announced plans to launch unbranded weight-loss drugs in Canada in January 2026 at discounts of up to 70% compared to branded versions.
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
