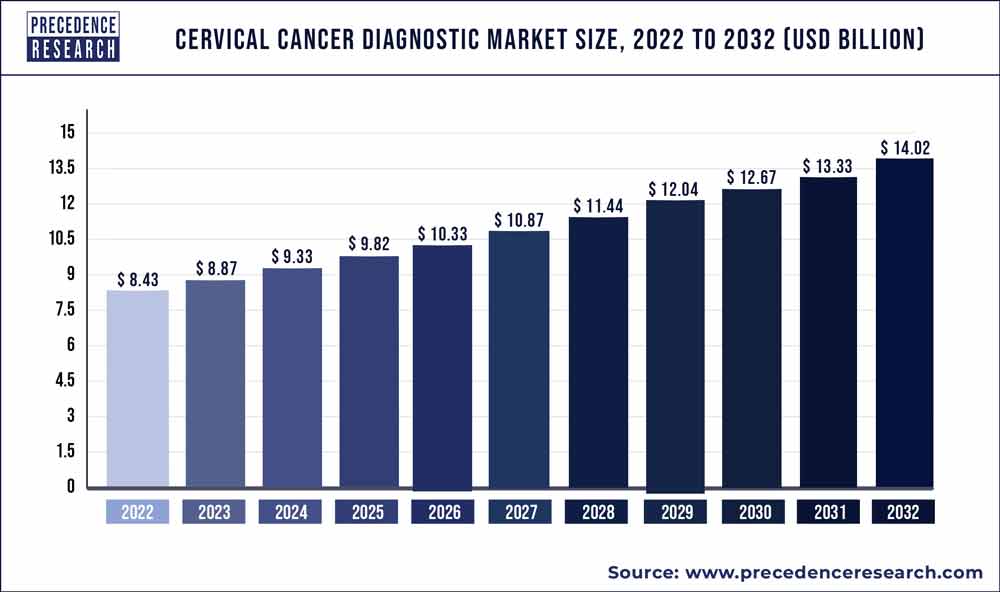
The global cervical cancer diagnostic market size was estimated to be around US$ 8.43 billion in 2022. It is projected to reach US$ 14.02 billion by 2032, indicating a CAGR of 5.22% from 2023 to 2032.
The market research report on the Cervical cancer diagnostic market provides a comprehensive analysis of various key aspects. It includes the definition, classification, and application of Cervical cancer diagnostic products. The report examines the development trends, competitive landscape, and industrial chain structure within the industry. Furthermore, it presents an overview of the industry, analyzes national policies and planning, and offers insights into the latest market dynamics and opportunities at a global level.
Get a Sample: https://www.precedenceresearch.com/sample/3178
Cervical Cancer Diagnostic Market Report Scope
| Report Coverage | Details |
| Market Size in 2023 | USD 8.87 Billion |
| Market Size by 2032 | USD 14.02 Billion |
| Growth Rate from 2023 to 2032 | CAGR of 5.22% |
| Largest Market | North America |
| Base Year | 2022 |
| Forecast Period | 2023 to 2032 |
| Segments Covered | By Diagnostic Test and By End-user |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Read More: Environmental Biotechnology Market Size, Share, Trends, Analysis Forecast By 2032
The report presents the volume and value-based market size for the base year 2022 and forecasts the market’s growth between 2023 and 2032. It estimates market numbers based on product form and application, providing size and forecast for each application segment in both global and regional markets.
Focusing on the global Cervical cancer diagnostic market, the report highlights its status, future forecasts, growth opportunities, key market players, and key market regions such as the United States, Europe, and China. The study aims to present the development of the Cervical cancer diagnostic market by considering factors like Year-on-Year (Y-o-Y) growth, in addition to Compound Annual Growth Rate (CAGR). This approach enables a better understanding of market certainty and the identification of lucrative opportunities.
Regarding production, the report investigates the capacity, production, value, ex-factory price, growth rate, and market share of major manufacturers, regions, and product types. On the consumption side, the report focuses on the regional consumption of Cervical cancer diagnostic products across different countries and applications.
Buyers of the report gain access to verified market figures, including global market size in terms of revenue and volume. The report provides reliable estimations and calculations for global revenue and volume by product type from 2023 to 2032. It also includes accurate figures for production capacity and production by region during the same period.
The research includes product parameters, production processes, cost structures, and data classified by region, technology, and application. Furthermore, it conducts SWOT analysis and investment feasibility studies for new projects.
This in-depth research report offers valuable insights into the Cervical cancer diagnostic market. It employs an objective and fair approach to analyze industry trends, supporting customer competition analysis, development planning, and investment decision-making. The project received support and assistance from technicians and marketing personnel across various links in the industry chain.
The competitive landscape section of the report provides detailed information on Cervical cancer diagnostic market competitors. It includes company overviews, financials, revenue generation, market potential, research and development investments, new market initiatives, global presence, production sites, production capacities, strengths and weaknesses, product launches, product range, and application dominance. However, the data points provided only focus on the companies’ activities related to the Cervical cancer diagnostic market.
Prominent players in the market are expected to face tough competition from new entrants. Key players are targeting acquisitions of startup companies to maintain their dominance. The report
Reasons to Purchase this Report:
- Comprehensive market segmentation analysis incorporating qualitative and quantitative research, considering the impact of economic and policy factors.
- In-depth regional and country-level analysis, examining the demand and supply dynamics that influence market growth.
- Market size in USD million and volume in million units provided for each segment and sub-segment.
- Detailed competitive landscape, including market share of major players, recent projects, and strategies implemented over the past five years.
- Comprehensive company profiles encompassing product offerings, key financial information, recent developments, SWOT analysis, and employed strategies by major market players.
Key Players
- Zilico
- Siemens Healthineers AG
- QIAGEN NV
- Abbott Laboratories
- Becton, Dickinson and Co.
- Quest Diagnostics Inc.
- F. Hoffmann-La Roche Ltd.
- Guided Therapeutics
- Hologic Inc.
- Bio-Rad Laboratories Inc.
Cervical Cancer Diagnostic Market Segmentations
By Diagnostic Test
- Pap Smear Test
- HPV Test
- Colposcopy
- Biopsy and Endocervical Curettage
- Other Diagnostic Tests
By End-user
- Hospitals
- Specialty Clinics
- Cancer and Radiation Therapy Centers
- Diagnostic Centers
- Others
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
TABLE OF CONTENT
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology (Premium Insights)
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Cervical Cancer Diagnostic Market
5.1. COVID-19 Landscape: Cervical Cancer Diagnostic Industry Impact
5.2. COVID 19 – Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Cervical Cancer Diagnostic Market, By Diagnostic Test
8.1. Cervical Cancer Diagnostic Market, by Diagnostic Test, 2023-2032
8.1.1. Pap Smear Test
8.1.1.1. Market Revenue and Forecast (2020-2032)
8.1.2. HPV Test
8.1.2.1. Market Revenue and Forecast (2020-2032)
8.1.3. Colposcopy
8.1.3.1. Market Revenue and Forecast (2020-2032)
8.1.4. Biopsy and Endocervical Curettage
8.1.4.1. Market Revenue and Forecast (2020-2032)
8.1.5. Other Diagnostic Tests
8.1.5.1. Market Revenue and Forecast (2020-2032)
Chapter 9. Global Cervical Cancer Diagnostic Market, By End-user
9.1. Cervical Cancer Diagnostic Market, by End-user, 2023-2032
9.1.1. Hospitals
9.1.1.1. Market Revenue and Forecast (2020-2032)
9.1.2. Specialty Clinics
9.1.2.1. Market Revenue and Forecast (2020-2032)
9.1.3. Cancer and Radiation Therapy Centers
9.1.3.1. Market Revenue and Forecast (2020-2032)
9.1.4. Diagnostic Centers
9.1.4.1. Market Revenue and Forecast (2020-2032)
9.1.5. Others
9.1.5.1. Market Revenue and Forecast (2020-2032)
Chapter 10. Global Cervical Cancer Diagnostic Market, Regional Estimates and Trend Forecast
10.1. North America
10.1.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.1.2. Market Revenue and Forecast, by End-user (2020-2032)
10.1.3. U.S.
10.1.3.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.1.3.2. Market Revenue and Forecast, by End-user (2020-2032)
10.1.4. Rest of North America
10.1.4.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.1.4.2. Market Revenue and Forecast, by End-user (2020-2032)
10.2. Europe
10.2.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.2.2. Market Revenue and Forecast, by End-user (2020-2032)
10.2.3. UK
10.2.3.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.2.3.2. Market Revenue and Forecast, by End-user (2020-2032)
10.2.4. Germany
10.2.4.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.2.4.2. Market Revenue and Forecast, by End-user (2020-2032)
10.2.5. France
10.2.5.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.2.5.2. Market Revenue and Forecast, by End-user (2020-2032)
10.2.6. Rest of Europe
10.2.6.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.2.6.2. Market Revenue and Forecast, by End-user (2020-2032)
10.3. APAC
10.3.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.3.2. Market Revenue and Forecast, by End-user (2020-2032)
10.3.3. India
10.3.3.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.3.3.2. Market Revenue and Forecast, by End-user (2020-2032)
10.3.4. China
10.3.4.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.3.4.2. Market Revenue and Forecast, by End-user (2020-2032)
10.3.5. Japan
10.3.5.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.3.5.2. Market Revenue and Forecast, by End-user (2020-2032)
10.3.6. Rest of APAC
10.3.6.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.3.6.2. Market Revenue and Forecast, by End-user (2020-2032)
10.4. MEA
10.4.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.4.2. Market Revenue and Forecast, by End-user (2020-2032)
10.4.3. GCC
10.4.3.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.4.3.2. Market Revenue and Forecast, by End-user (2020-2032)
10.4.4. North Africa
10.4.4.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.4.4.2. Market Revenue and Forecast, by End-user (2020-2032)
10.4.5. South Africa
10.4.5.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.4.5.2. Market Revenue and Forecast, by End-user (2020-2032)
10.4.6. Rest of MEA
10.4.6.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.4.6.2. Market Revenue and Forecast, by End-user (2020-2032)
10.5. Latin America
10.5.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.5.2. Market Revenue and Forecast, by End-user (2020-2032)
10.5.3. Brazil
10.5.3.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.5.3.2. Market Revenue and Forecast, by End-user (2020-2032)
10.5.4. Rest of LATAM
10.5.4.1. Market Revenue and Forecast, by Diagnostic Test (2020-2032)
10.5.4.2. Market Revenue and Forecast, by End-user (2020-2032)
Chapter 11. Company Profiles
11.1. Zilico
11.1.1. Company Overview
11.1.2. Product Offerings
11.1.3. Financial Performance
11.1.4. Recent Initiatives
11.2. Siemens Healthineers AG
11.2.1. Company Overview
11.2.2. Product Offerings
11.2.3. Financial Performance
11.2.4. Recent Initiatives
11.3. QIAGEN NV
11.3.1. Company Overview
11.3.2. Product Offerings
11.3.3. Financial Performance
11.3.4. Recent Initiatives
11.4. Abbott Laboratories
11.4.1. Company Overview
11.4.2. Product Offerings
11.4.3. Financial Performance
11.4.4. Recent Initiatives
11.5. Becton, Dickinson and Co.
11.5.1. Company Overview
11.5.2. Product Offerings
11.5.3. Financial Performance
11.5.4. Recent Initiatives
11.6. Quest Diagnostics Inc.
11.6.1. Company Overview
11.6.2. Product Offerings
11.6.3. Financial Performance
11.6.4. Recent Initiatives
11.7. F. Hoffmann-La Roche Ltd.
11.7.1. Company Overview
11.7.2. Product Offerings
11.7.3. Financial Performance
11.7.4. Recent Initiatives
11.8. Guided Therapeutics
11.8.1. Company Overview
11.8.2. Product Offerings
11.8.3. Financial Performance
11.8.4. Recent Initiatives
11.9. Hologic Inc.
11.9.1. Company Overview
11.9.2. Product Offerings
11.9.3. Financial Performance
11.9.4. Recent Initiatives
11.10. Bio-Rad Laboratories Inc.
11.10.1. Company Overview
11.10.2. Product Offerings
11.10.3. Financial Performance
11.10.4. Recent Initiatives
Chapter 12. Research Methodology
12.1. Primary Research
12.2. Secondary Research
12.3. Assumptions
Chapter 13. Appendix
13.1. About Us
13.2. Glossary of Terms
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com