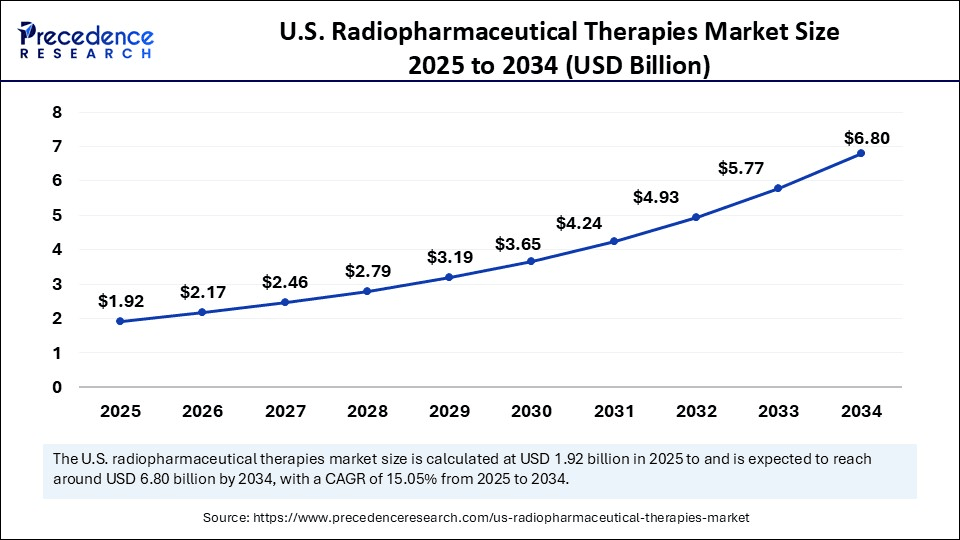
U.S. Radiopharmaceutical Therapies Market Key Takeaways
- By therapy type, the targeted beta therapy segment held the largest share of 66.29% in 2024.
- By therapy type, the targeted alpha therapy segment is expected to grow at the fastest CAGR of 25.27% during the forecast period.
- By radioisotopes used, the beta emitters segment held the largest share of 66.29% in 2024.
- By radioisotopes used, the alpha emitters segment is expected to grow at the fastest CAGR of 25.26% in the upcoming period.
- By therapeutic area, the oncology segment held the biggest market share of 88.14% in 2024.
- By therapeutic area, the non-oncology segment is emerging as the fastest growing during the forecast period.
- By route of administration, the intravenous injection segment generated the major market share of 80.14% in 2024.
- By route of administration, the intratumoral (investigational) segment is expected to grow at the fastest CAGR of 25.06% during the forecast period.
- By end-user, the hospitals segment held the highest market share of 62.81% in 2024.
- By end-user, the radiopharmacies (centralized radiopharmacy networks) segment is observed to grow at the fastest CAGR of 17.50% during the forecast period.
Impact of AI on the U.S. Radiopharmaceutical Therapies Market
Artificial Intelligence is transforming the U.S. radiopharmaceutical therapies market by enhancing diagnostic precision, streamlining drug development, and enabling personalized treatment approaches. In nuclear medicine, AI-powered imaging tools improve the detection of cancer lesions, supporting more accurate targeting of therapies like Pluvicto and Lutathera. This leads to better patient selection and improved clinical outcomes.
AI also enhances PET/CT scan accuracy, supports tailored dosimetry for safer treatments, and expedites drug discovery by modeling ligand-receptor interactions. Additionally, it optimizes the logistics of delivering time-sensitive isotopes. As AI becomes more integrated into nuclear medicine workflows, it is shaping a more efficient, individualized, and scalable future for radiopharmaceutical care in the U.S.
Market Overview
The U.S. radiopharmaceutical therapies market is experiencing robust expansion, driven by escalating demand for precision medicine in oncology and nuclear diagnostics. In 2024 alone, over 2 million new cancer cases increased reliance on targeted radioligand therapies and PET‐based diagnostics. This sector is evolving rapidly as theranostics—combining therapeutic and diagnostic functions in unified platforms—become standard practice. Market forecasts predict high double‑digit year‑on‑year growth through the 2020s, underpinned by new FDA approvals of isotopic agents and expanding use of PET/SPECT technologies in major clinical settings.
Market Drivers
Cancer and chronic disease prevalence: The U.S. aging population (65+ demographic) is more prone to cancer, neurodegenerative diseases, and cardiovascular conditions. Radiopharmaceuticals offer the high sensitivity and specificity needed for early disease detection and targeted treatment.
Technological innovation: Advances in isotope production (gallium‑68, lutetium‑177, actinium‑225) and automated synthesizers are enhancing both the availability and precision of therapies and diagnostics. Integration of AI in imaging protocols and development pipelines accelerates drug discovery and improves accuracy in PET/SPECT interpretation.
Regulatory and reimbursement support: The FDA has cleared multiple new agents for prostate cancer, neuroendocrine tumors, and brain imaging. Medicare proposals to improve reimbursement for high‑cost diagnostics (e.g. Lantheus’ agents) are boosting provider uptake by easing financial uncertainties.
Opportunities
Precision oncology and personalized theranostics: Theranostic platforms are emerging as a core growth driver, particularly in prostate, neuroendocrine, and metastatic cancers. Pierceps like Lutetium‑177‑PSMA therapies offer dual diagnostic and therapeutic applications.
Decentralized production: Mobile radiopharmacy units and localized cyclotron hubs allow wider access in rural and underserved areas, mitigating distribution constraints from short isotope half‑lives. Investment in domestic isotope supply (e.g. actinium‑225) enhances control and scalability.
AI‑driven development and decision‑support: AI and machine learning are optimizing not only imaging workflows but also dosimetry and compound discovery—reducing time‑to‑market and improving patient outcomes.
Challenges
Complex supply chain and short half‑lives: Many therapeutic isotopes degrade rapidly, necessitating tightly coordinated logistics between production, labeling, and clinical administration. Reliance on a limited number of cyclotron or reactor facilities increases vulnerability to disruptions.
High production and infrastructure costs: Building cyclotron suites, maintaining GMP standards, and scaling isotope synthesis come with major capital expenditures. These costs often restrict access and limit entry for smaller players.
Regulatory complexity and reimbursement uncertainty: Approval pathways remain lengthy, and inconsistent reimbursement policies across private payers and government programs create financial ambiguity for providers adopting new radiopharmaceutical technologies.
Recent Developments
In early 2025, a leading pharmaceutical company acquired a U.S. radiotracer manufacturer to expand PET imaging capacity. Academic‑industry consortiums launched collaborative R&D hubs to commercialize university‑developed agents, improving domestic self‑sufficiency.
Microsoft and Telix (through Subtle Medical collaboration) deployed an AI‑enhanced PSMA‑PET workflow called SubtlePET, cutting scan duration by up to 75% while preserving image quality—sparking improved workflow efficiency in imaging centers.
Private investors and analysts remain bullish: Novartis’ Pluvicto exceeded $1 billion in sales through late 2024, prompting acquisitions by AstraZeneca, Eli Lilly, and Bristol‑Myers Squibb of radiopharma innovators and startups. Eli Lilly also invested $10 million in Ionetix to bolster domestic actinium‑225 production.
Get Free sample @ https://www.precedenceresearch.com/sample/6508
U.S. Radiopharmaceutical Therapies Market Companies
- Bayer
- Novartis
- China Isotope & Radiation
- Dongcheng
- Q BioMed
- Curium Pharmaceuticals
- Jubilant DraxImage
- Lantheus
- Spectrum Pharmaceuticals
- Progenics Pharmaceuticals
- International Isotopes
Segments Covered in the Report
By Therapy Type
- Targeted Alpha Therapy (TAT)
- Actinium-225-based therapies
- Radium-223-based therapies
- Astatine-211-based therapies
- Targeted Beta Therapy
- Lutetium-177-based therapies (e.g., Lu-177 DOTATATE)
- Iodine-131-based therapies
- Yttrium-90-based therapies
- Brachytherapy-based Radiopharmaceuticals
- Auger Electron Emitting Radiopharmaceuticals (emerging)
- Others
By Radioisotope Used
- Beta Emitters
- Lutetium-177 (Lu-177)
- Iodine-131 (I-131)
- Yttrium-90 (Y-90)
- Alpha Emitters
- Actinium-225 (Ac-225)
- Radium-223 (Ra-223)
- Astatine-211 (At-211)
- Brachytherapy-based Radiopharmaceuticals
- Auger Electron Emitting Radiopharmaceuticals (emerging)
- Others
- Samarium-153
- Rhenium-186/188
- Holmium-166
By Therapeutic Area
- Oncology
- Prostate cancer
- Neuroendocrine tumors
- Liver cancer (e.g., HCC)
- Thyroid cancer
- Non-Hodgkin’s lymphoma
- Non-Oncology
- Bone metastases pain palliation
- Hyperthyroidism
- Cardiology (e.g., atrial fibrillation ablation using radioisotopes)
By Route of Administration
- Intravenous Injection
- Oral (primarily Iodine-131)
- Intra-arterial (in case of liver cancer, radioembolization)
- Intratumoral (investigational)
By End-User
- Hospitals
- Academic Medical Centers
- Specialty Cancer Hospitals
- Radiopharmacies (Centralized Radiopharmacy Networks)
- Ambulatory Surgical Centers (ASCs)
- Nuclear Medicine Centers
Also Visit @https://www.precedenceresearch.com/
