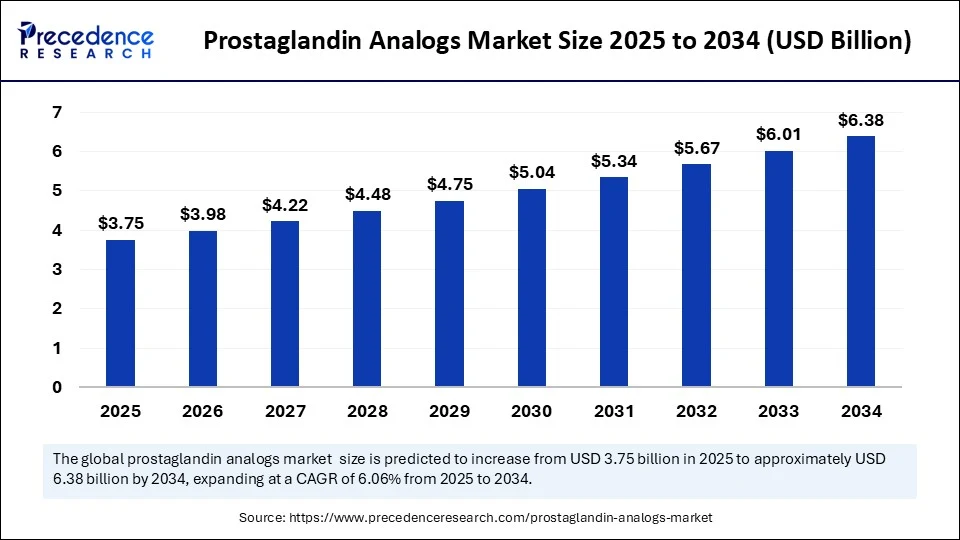
Market Overview
The Prostaglandin Analogs Market plays a vital role in the treatment of glaucoma and ocular hypertension by reducing intraocular pressure. As a key segment within ophthalmic pharmaceuticals, the Prostaglandin Analog Drugs Market has seen consistent growth due to increasing prevalence of eye disorders worldwide. This market comprises several leading products designed to enhance patient outcomes while minimizing side effects, supported by expanding healthcare infrastructure and awareness campaigns.
AI and Innovation
Artificial intelligence is increasingly influencing the Prostaglandin Analogs Market by optimizing drug development and personalized treatment regimens. In the Prostaglandin Analog Drugs Market, AI-powered platforms assist in identifying patient-specific responses and adverse effect profiles, enhancing therapy effectiveness. Innovation also drives novel delivery systems such as sustained-release implants and nanotechnology-based formulations, improving drug bioavailability and patient compliance.
Future Trends of Market
Future trends in the Prostaglandin Analogs Market indicate rising adoption of combination therapies and targeted delivery mechanisms. The Prostaglandin Analog Drugs Market is expected to embrace advancements such as gene therapy and regenerative medicine approaches to treat glaucoma more effectively. Additionally, increasing investments in R&D are fostering the development of next-generation analogs with enhanced efficacy and safety profiles.
Rising Demands of Market
Rising demand in the Prostaglandin Analogs Market is driven by the growing incidence of glaucoma and age-related eye conditions. The Prostaglandin Analog Drugs Market benefits from greater patient awareness and improvements in diagnostic techniques that enable early detection. As global populations age, the need for effective, well-tolerated treatment options continues to expand, supporting steady market growth.
Key Market Highlights
Key highlights within the Prostaglandin Analogs Market include recent regulatory approvals for innovative formulations and strategic alliances among pharmaceutical companies. The Prostaglandin Analog Drugs Market has also witnessed the launch of generic versions, making treatments more accessible and affordable. Market leaders are focusing on expanding distribution networks and enhancing patient education to maximize product reach.
Market Growth Drivers
The Prostaglandin Analogs Market growth is driven by increasing prevalence of ocular diseases, technological advancements, and supportive healthcare policies. The Prostaglandin Analog Drugs Market also benefits from rising healthcare expenditure and insurance coverage expansion. Growing emphasis on early diagnosis and treatment adherence further fuels demand for prostaglandin analog therapies globally.
Restraints
Despite growth, the Prostaglandin Analogs Market faces challenges such as side effects including ocular irritation and hyperemia, which may limit patient compliance. The Prostaglandin Analog Drugs Market is also restrained by patent expirations leading to pricing pressures and competition from alternative drug classes. Additionally, stringent regulatory requirements and high R&D costs pose barriers to new product introductions.
Opportunities
Opportunities in the Prostaglandin Analogs Market include expansion into emerging markets where glaucoma prevalence is rising alongside improving healthcare infrastructure. The Prostaglandin Analog Drugs Market can capitalize on technological innovations like sustained-release delivery and personalized medicine. Increasing collaborations between biotech firms and pharmaceutical companies offer promising avenues for product development.
Also Read@ https://www.dailytechbulletin.com/semiconductor-wafer-transfer-robots-market/
Prostaglandin Analogs Market Companies

- Alcon (Novartis division)
- Allergan (AbbVie) (Lumigan – Bimatoprost)
- Bausch + Lomb (part of Bausch Health)
- Bausch Health Companies Inc.
- Chengdu Kanghong Pharmaceutical Group Co., Ltd.
- Dr. Reddy’s Laboratories Ltd.
- Eagle Pharmaceuticals, Inc.
- Fresenius Kabi AG
- Glenmark Pharmaceuticals Ltd.
- Hugel, Inc.
- Ipsen
- Macleods Pharmaceuticals Ltd.
- Mylan N.V. (Viatris)
- Novartis AG
- Pfizer Inc. (Xalatan – Latanoprost)
- Santen Pharmaceutical Co., Ltd.
- STADA Arzneimittel AG
- Sun Pharmaceutical Industries Ltd.
- Sunovion Pharmaceuticals Inc.
- Zhejiang Xianju Pharmaceutical Co., Ltd.
Recent Developments
- In January 2025, Glaukos Corporation (NYSE: GKOS), an ophthalmic pharmaceutical and medical technology company specializing in therapies for glaucoma, corneal disorders, and retinal diseases, announced several positive clinical updates for its iDose sustained-release procedural pharmaceutical platform.
- In December 2024, the U.S. Food and Drug Administration (FDA) approved a generic version of latanoprost, a prostaglandin analog, for treating open-angle glaucoma and ocular hypertension. According to Gland Pharma Limited, the FDA determined that the generic product is both bioequivalent and therapeutically equivalent to Xalatan (latanoprost 0.005%, Pfizer).
- In December 2023, the FDA approved the iDose TR sustained-release implant, a prostaglandin analog delivery device that continuously administers 75 mcg of travoprost for up to 36 months. This single-administration implant helps lower intraocular pressure (IOP) and could reduce or eliminate the need for daily topical drops. Given the challenges of patient compliance with topical therapies, this development provides ophthalmologists with a long-acting alternative to manage ocular hypertension and open-angle glaucoma.
- In June 2025, Qlaris Bio, a clinical-stage biotechnical company, announced the development of a novel preservative-free, fixed-dose combination therapy (QLS-111-FDC) combining QLS-111 and latanoprost. The therapy targets primary open-angle glaucoma (POAG), ocular hypertension (OHT), and normal-tension glaucoma (NTG), particularly for patients needing additional IOP reduction via episcleral venous pressure (EVP) modulation. QLS-111 addresses EVP, a component of IOP not targeted by current approved treatments, representing a potential advancement in glaucoma management.
Get Sample link @ https://www.precedenceresearch.com/sample/6595
