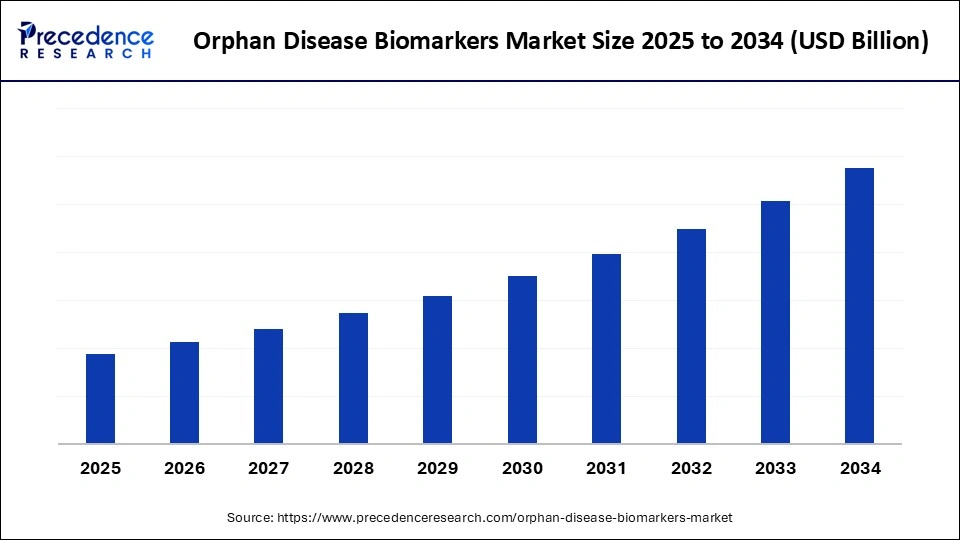
. Market Overview
The Orphan Disease Biomarkers Market is undergoing significant transformation as the demand for precision diagnostics in rare diseases intensifies. Rare conditions, often lacking effective treatments, are now at the forefront of biomarker research. The market has seen rapid evolution, driven by growing awareness, earlier diagnostic needs, and targeted therapy development.
Biomarkers help clinicians detect disease progression, determine prognosis, and monitor treatment efficacy. In rare diseases, where traditional diagnostic methods fall short, these biomarkers are essential. As research institutions and biotech companies increase their focus on rare disorders, the Orphan Disease Biomarkers Market continues to expand and mature.
2. AI and Innovation in Biomarker Discovery
Technological innovation is accelerating growth in the Orphan Disease Biomarkers Market. Artificial Intelligence (AI) and machine learning are redefining biomarker discovery. Algorithms now sift through vast omics datasets to identify candidate biomarkers in rare disease populations.
AI enhances accuracy in pattern recognition, especially in genomics and proteomics, where rare conditions leave subtle molecular traces. Platforms leveraging deep learning enable real-time predictions, reducing trial-and-error in validation processes. This wave of digital innovation is shortening development timelines and increasing the success rate of clinical translation.
3. Future Trends in the Market
The future of the Orphan Disease Biomarkers Market is shaped by a growing shift toward multi-omics integration, regulatory incentives for rare disease research, and decentralized diagnostics. Researchers are combining genomics, transcriptomics, metabolomics, and epigenomics to build comprehensive biomarker profiles.
Another emerging trend is the focus on pediatric populations. Many orphan diseases are congenital, requiring early-life diagnosis. Furthermore, the integration of wearable health devices and biosensors is pushing real-time biomarker tracking from research labs to home care environments.
4. Rising Demands of the Market
As the prevalence of rare diseases becomes more visible globally, the Orphan Disease Biomarkers Market faces rising demand from both researchers and healthcare providers. Advocacy from patient groups and government funding has accelerated awareness and investments.
Pharmaceutical companies now prioritize rare disease segments due to market exclusivity and expedited regulatory pathways. With over 7,000 rare diseases affecting millions, the need for accurate, early-stage biomarkers continues to grow rapidly. This surging demand fuels the ongoing development of personalized diagnostics and companion tests.
5. Key Market Highlights
-
Growing adoption of multi-omics platforms.
-
Increase in rare disease-specific biomarker pipelines.
-
AI-assisted discovery tools gain commercial traction.
-
Pediatric diagnostics for rare conditions rise in importance.
-
Clinical trials are incorporating biomarker-based endpoints more frequently.
These highlights illustrate the expanding scope of the Orphan Disease Biomarkers Market and its increasing role in modern healthcare.
6. Market Growth Drivers
The Orphan Disease Biomarkers Market is driven by several critical factors:
-
Technological Advancement: Enhanced bioinformatics tools and sequencing platforms.
-
Regulatory Support: Orphan drug incentives encourage diagnostic development.
-
R&D Funding: Global increase in public and private investment.
-
Unmet Medical Needs: The absence of approved diagnostics for thousands of conditions.
-
Pharma Interest: Rise in rare disease drug pipelines requiring companion diagnostics.
Each of these forces works synergistically to strengthen the market’s trajectory.
7. Market Restraints
Despite growth, the Orphan Disease Biomarkers Market faces notable restraints:
-
Limited Patient Pools: Small sample sizes complicate validation efforts.
-
High R&D Costs: Multi-omics platforms and clinical trials are expensive.
-
Regulatory Complexity: Approval pathways can be inconsistent across regions.
-
Data Privacy: Genomic data raises ethical concerns in biomarker research.
Addressing these challenges will be essential for sustained progress.
8. Market Opportunities
The market presents compelling opportunities for stakeholders:
-
Rare Neurological Disorders: Biomarkers for ALS, Huntington’s, and Batten disease.
-
Liquid Biopsy Expansion: Non-invasive diagnostics tailored for rare cancers.
-
Decentralized Trials: Biomarker-based home testing and monitoring.
-
AI-driven Platforms: Tools focused specifically on rare and ultra-rare disease detection.
These opportunities highlight the untapped potential in the Orphan Disease Biomarkers Market.
9. Regional Insights
-
North America leads with strong R&D infrastructure, orphan drug laws, and advanced diagnostics networks.
-
Europe follows closely, with a focus on collaborative research and government-funded rare disease initiatives.
-
Asia-Pacific is emerging fast, driven by increasing awareness and genomics investment in countries like China, Japan, and South Korea.
-
Latin America & Middle East are nascent markets showing potential due to improved healthcare systems and regional research collaborations.
Orphan Disease Biomarkers Market Companies

- Roche
- Thermo Fisher Scientific
- Abbott Laboratories
- QIAGEN
- Bio-Rad Laboratories
- PerkinElmer
- Illumina
- Agilent Technologies
- Merck KGaA
- Siemens Healthineers
- GE Healthcare
- Danaher Corporation
- Novartis
- BioMérieux
- Abbott Diagnostics
- Medtronic
- F. Hoffmann-La Roche AG
- Labcorp Drug Development
- Invitae Corporation
- Eurofins Scientific
Recent Developments
- In July 2025, NIH issued a Request for Applications (RFA) calling for biomarker and clinical outcome assessment development for rare diseases, to be conducted alongside prospective natural history studies—supporting rigorous trial design.
- In July 2025, Alterity Therapeutics reported positive topline results from its Phase 2 trial of ATH434 in Multiple System Atrophy (MSA), and earlier in May secured Fast Track Designation for the same asset—indicating strong clinical momentum
- In April 2025, U.S. FDA Commissioner Marty Makary announced plans for a new ultrarare drug approval pathway based on a “plausible mechanism,” potentially enabling conditional approvals without randomized, controlled trials.
Get Free Sample Link @ https://www.precedenceresearch.com/sample/6756
