Inactivated Vaccines Market Key Takeaways
- In terms of revenue, the global inactivated vaccines market was valued at USD 42.97 billion in 2024.
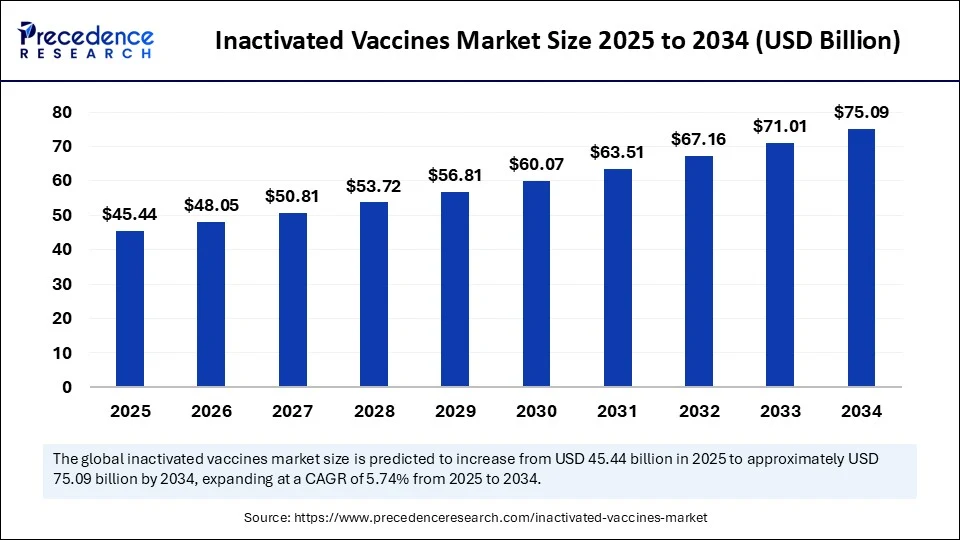
- It is projected to reach USD 75.09 billion by 2034.
- The market is expected to grow at a CAGR of 5.74% from 2025 to 2034.
- Asia Pacific dominated the inactivated vaccines market with the largest share of 39% in 2024.
- The Middle East & Africa is expected to grow at a significant CAGR from 2025 to 2034.
- By vaccine type, the whole virus inactivated vaccines segment held the major market share of 46% in 2024.
- By vaccine type, the subunit inactivated vaccines segment is projected to grow at the highest CAGR between 2025 and 2034.
- By disease indication, the polio segment captured the biggest market share of 27% in 2024.
- By disease indication, the COVID-19 segment is likely to expand at a significant CAGR from 2025 to 2034.
- By age group, the pediatrics (0–18 years) segment contributed the largest market share of 63% in 2024.
By age group, the geriatrics segment is anticipated to grow at a significant CAGR from 2025 to 2034. - By end user, the public immunization programs segment generated the major market share of 68% in 2024.
- By end user, the travel & occupational health centers segment is expected to grow at a significant CAGR over the projected period.
- By distribution channel, the government supply contracts segment led the market in 2024.
- By distribution channel, the direct institutional sales segment is expected to grow at a significant CAGR from 2025 to 2034.
How is AI Impacting the Inactivated Vaccines Market?
Artificial intelligence (AI) is reshaping the inactivated vaccines market by accelerating development timelines and enhancing overall efficiency. From streamlining antigen discovery to optimizing vaccine design and improving the accuracy of preclinical and clinical testing, AI significantly boosts productivity across the development pipeline. AI-driven models simulate immune responses to antigens, helping design immunogens that elicit strong and targeted immune reactions.
In clinical trials, AI supports faster patient recruitment, predicts potential adverse events, and enables adaptive protocols that adjust in real time—reducing trial duration and speeding up regulatory approval. Once deployed, AI continues to add value by monitoring vaccine safety and effectiveness through real-world data sources, including electronic health records, social media, and adverse event databases.
Market Overview
The inactivated vaccines market—comprising vaccines where pathogens are rendered non‑replicative through heat, chemicals, or radiation—is a mature but dynamically evolving segment of the global immunization ecosystem. In the U.S., Europe, and emerging economies, vaccines against influenza, polio, hepatitis A, rabies, and certain COVID‑19 formulations remain mainstay therapies.
With global demand recovering post‑pandemic and routine immunization programs expanding, the market enjoys steady growth. Emerging technologies in antigen stabilization, adjuvant formulations, and large‑scale bioreactor production are enhancing yield, shelf‑life, and immunogenicity, translating into robust growth in both public and private vaccine procurement channels.
Market Drivers
Widespread established use and trust: Inactivated vaccines enjoy long-standing regulatory approval, high safety profiles, and public acceptance globally. Many national immunization programs rely on them for children and adult boosters. Pandemic recovery and preparedness: The COVID‑19 pandemic spotlighted the need for scalable, proven platforms; inactivated vaccine lines are being scaled to provide backup options to mRNA and vector approaches.
Technological refinement: Innovations in viral inactivation processes, micro‑encapsulation adjuvants, controlled antigen release systems, and lyophilization methods are enhancing stability and dose-sparing—also enabling improved cold‑chain resilience. Government and NGO funding: Global health agencies continue to allocate resources for vaccine stockpiling, outbreak preparedness, and expanded routine coverage, reinforcing demand for established inactivated platforms.
Opportunities
Emerging market penetration: Low‑ and middle‑income countries are accelerating adoption of affordable inactivated vaccine variants, particularly for influenza, hepatitis A and polio. Co‑formulated inactivated vaccines that protect against multiple pathogens in a single shot offer promising value and simplified logistics. Dose‑sparing and next‑generation adjuvants: Research into novel immune enhancers (e.g., nanoparticle adjuvants, T‑cell stimulators) can significantly reduce antigen dosage per vial, lowering costs and expanding supply capacity.
Stockpiling for pandemics and bio‑threats: Governments and global agencies are establishing vaccine reserves for influenza and other potential threats; inactivated platforms—owing to proven infrastructure—are preferred for rapid scale-up and rollout. Improved delivery technologies: Microneedle patches, inhalable dry powder formulations, and controlled‑release vehicles can reduce cold-chain reliance and injection requirements, making vaccine delivery more efficient and accessible.
Challenges
Competition from novel platforms: mRNA and viral‑vector vaccines continue to attract attention for rapid development and robust immunogenicity. Inactivated vaccines—while proven—can appear slower to develop and require traditional cold‑chain infrastructure, potentially reducing attractiveness in fast-moving outbreak scenarios.
Manufacturing complexity: Growing large quantities of infectious virus safely, followed by validated inactivation and purification, demands high biosafety levels, stringent quality controls, and specialized facilities. This raises upfront capital costs, limits scale-up speed, and may restrict new entrants. Cold‑chain dependency: While lyophilization helps, many inactivated vaccines still require refrigerated storage and transport, posing challenges in remote or low-resource areas. Regulatory and batch release timelines: Though regulatory pathways are well-established, batch testing, sterility and potency validation can be time‑consuming, impacting response time during emergent situations.
Recent Developments
In the latest period, several vaccine manufacturers have expanded inactivated influenza production to support growing adult and pediatric booster demand. Some firms have introduced quadrivalent inactivated flu vaccines with enhanced antigen-adjuvant combinations to extend seasonal coverage and immune memory duration.
Inactivated COVID‑19 vaccine programs in Asia and Latin America have scaled operations and begun export to secondary markets, offering bridged formulations with high thermostability. Novel microneedle patch trials are underway using inactivated hepatitis A antigens, successfully demonstrating dose-sparing and self‑administration potential.
Global health agencies are funding multi‑dose inactivated polio and enterovirus vaccine stockpiles to pre‑empt future outbreaks. Simultaneously, manufacturers are optimizing continuous viral culture and inactivation platforms using single-use bioreactors and automated purification workflows to reduce contamination risk and increase batch throughput.
Get Sample Link@ https://www.precedenceresearch.com/sample/6506
Inactivated Vaccines Market Companies
- Sanofi Pasteur
- GSK plc
- Bharat Biotech (Covaxin, Typbar)
- Sinovac Biotech Ltd.
- Sinopharm (CNBG)
- Serum Institute of India (SII)
- Pfizer Inc.
- Valneva SE
- Moderna, Inc. (expanding into inactivated pipeline)
- Biological E. Limited
- Panacea Biotec Ltd.
- Chumakov Institute of Poliomyelitis and Viral Encephalitides (Russia)
- IDT Biologika
- Emergent BioSolutions
- Novavax, Inc. (subunit but related pipeline)
- Incepta Vaccine Ltd. (Bangladesh)
- Haffkine Bio-Pharmaceutical Corporation Ltd.
- Bio Farma (Indonesia)
- Instituto Butantan (Brazil)
- Walvax Biotechnology Co., Ltd. (China)
Segments Covered in the Report
By Vaccine Type
- Whole Virus Inactivated Vaccines
- Inactivated Polio Vaccine (IPV)
- Rabies Vaccine
- Hepatitis A Vaccine
- Inactivated COVID-19 Vaccines (e.g., Sinovac, Covaxin)
- Split-Virion Inactivated Vaccines
- Subunit Inactivated Vaccines
- Pertussis (aP)
- Influenza (Split Virion, Subunit)
- Tetanus & Diphtheria
- Combination Inactivated Vaccines
- DTP
- DTaP-IPV-HepB-Hib
- Adjuvanted Inactivated Vaccines
By Disease Indication
- Polio
- Influenza
- COVID-19
- Hepatitis A
- Rabies
- Japanese Encephalitis
- Cholera
- Pertussis
- Tick-Borne Encephalitis
- Combination Indications
By Age Group
- Pediatrics (0–18 years)
- Adults
- Geriatrics
- Travelers & At-Risk Populations
By End User
- Public Immunization Programs
- Private Clinics & Hospitals
- Military & Emergency Preparedness
- Travel & Occupational Health Centers
By Distribution Channel
- Government Supply Contracts (e.g., UNICEF, Gavi, WHO)
- Hospital Pharmacies
- Retail Pharmacies
- Direct Institutional Sales
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Also Visit@ https://www.precedenceresearch.com/
