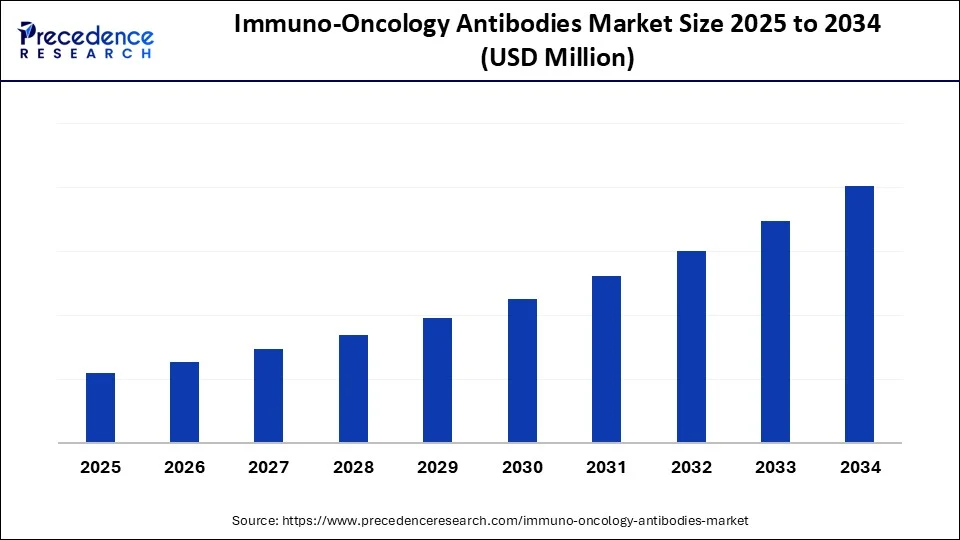
Immuno-Oncology Antibodies Market Key Takeaways
- North America dominated the immuno-oncology antibodies market with the largest share of 48.7% in 2024.
- Asia Pacific is expected to grow at the fastest CAGR from 2025 to 2034.
- By drug class, the checkpoint inhibitors segment led the market in 2024, under which the PD-1 inhibitors hold a 32.1% as a sub-segment.
- By drug class, the bispecific antibodies segment is expected to grow at the fastest CAGR in the upcoming period.
- By application, the non-small cell lung cancer (NSCLC) segment captured the biggest market share of 28.4% in 2024.
- By application, the triple-negative breast cancer (TNBC) segment is expected to grow at a significant CAGR over the projected period.
- By mechanism of action, the immune checkpoint blockade segment contributed the biggest market share of 45.7% in 2024.
- By mechanism of action, the immune cell redirection segment is anticipated to grow at a significant CAGR from 2025 to 2034.
- By route of administration, the intravenous (IV) segment held the highest market share of 81.3% in 2024.
- By route of administration, the subcutaneous (SC) segment is expanding at a significant CAGR from 2025 to 2034.
- By end user, the hospitals segment held the major market share of 61.2% in 2024.
- By end user, the cancer specialty clinics segment is projected to grow at a significant CAGR between 2025 and 2034.
- By distribution channel, the hospital pharmacies segment generated the largest market share of 52.6% in 2024.
- By distribution channel, the specialty pharmacies segment is expected to experience rapid growth during the forecast period.
How Can AI Impact the Immuno-Oncology Antibodies Market?
In the Immuno-Oncology Antibodies Market, AI technologies have advanced rapidly: foundation models now predict antibody–antigen binding and functional properties with state‑of‑the‑art AUROC scores (~0.91 for known antigens, ~0.73 for unseen).
Generative humanization models produce candidate sequences that are highly human-like while preserving therapeutic potential. Biotech Venture studios such as AION Labs, backed by pharma and tech giants, incubated DenovAI and CombinAble.AI to drive antibody design via AI. These infrastructures promote more efficient candidate generation and reduced discovery timelines.
Market Overview
The Immuno-Oncology Antibodies Market combines several therapeutic formats—including checkpoint monoclonals, bispecific T‑cell engagers, masked constructs, and ADCs—to harness immune mechanisms against cancer. Over the last decade, therapies like pembrolizumab and nivolumab have transformed oncology; meanwhile, the innovation frontier now centers on multispecific and tumor‑selective antibodies.
The market is supported by increasing clinical adoption, intensifying M&A and licensing activity, and growing acceptance of immunotherapy in standard oncology protocols.
Drivers
Catalysts for growth in the Immuno-Oncology Antibodies Market:
-
Continued global rise in cancer rates (especially lung, colorectal, melanoma).
-
Proven efficacy and durability of checkpoint inhibitors propelling first‑line adoption.
-
Emergence of bispecific, trispecific, and ADC formats enhancing potency and selectivity.
-
Heavy industry investment and expedited regulatory pathways for novel immunotherapies.
-
Broadening reimbursement coverage in developed markets enabling greater patient access.
Market Trends
Current trends include:
-
Expansion of bispecific and multispecific antibody drugs targeting immune and tumor pathways.
-
Rapid growth in ADC platforms, expected to rival traditional chemotherapy in certain cancers.
-
Integration of AI/ML methods in trial design, biomarker discovery, and candidate screening.
-
Increasing use of combination regimens involving IO antibodies, vaccines, CAR‑T, and checkpoint inhibitors.
-
Dealmaking surge in the ADC and multispecific sectors, with acquisitions and licensing totaling billions annually.
Opportunities
Potential growth avenues within the Immuno-Oncology Antibodies Market:
-
Development and commercialization of next‑generation bispecific therapies that may outperform immune checkpoint monotherapies.
-
Geographical expansion into emerging economies via evolving healthcare systems and growing awareness.
-
Use of AI discovery platforms to democratize access to novel antibody design for smaller biotech firms.
-
Strategic collaborations to fast-track pipeline assets, such as the Bristol Myers‑BioNTech partnership on BNT327.
-
Biomarker‑driven personalization and adaptive trial designs that optimize outcomes and expand indications.
Challenges
Ongoing challenges include:
-
High cost of drug development and therapy limits global access and presents reimbursement strain.
-
Regulatory scrutiny for novel antibody modalities requires extensive validation.
-
Safety management, particularly of immune‑related toxicities and CRS.
-
AI bias and data limitations in antibody discovery may undermine generalizability.
-
Patent expirations and biosimilar entry threaten legacy monoclonal antibody revenue.
-
Competitive intensity necessitates constant innovation among large and emerging firms.
Also read@ https://www.dailytechbulletin.com/liposomal-supplements-market
Recent Developments
Recent highlights shaping the Immuno-Oncology Antibodies Market:
-
Cosibelimab‑ipdl was FDA-approved in late 2024 for advanced cutaneous squamous cell carcinoma.
-
The Bristol Myers‑BioNTech deal in June 2025 commits up to USD 11.1 billion to develop BNT327, a PD‑L1/VEGF bispecific antibody.
-
Generate:Biomedicines initiated a high‑value collaboration with Novartis in September 2024 to employ its AI platform in antibody design.
-
AION Labs launched CombinAble.AI in early 2025 to generate multispecific biologics more efficiently.
-
Agenus restructured by exiting CDMO operations and forming commercialization partnerships in India to focus on pipeline advancement.
-
Genmab acquired ProfoundBio in early 2024 for USD 1.8 billion, gaining access to novel ADC candidates including Rina‑S (fast‑track designation for ovarian cancer).
Immuno-Oncology Antibodies Market Companies
- Bristol Myers Squibb
- Merck & Co., Inc.
- Roche Holding AG
- AstraZeneca
- Pfizer Inc.
- Regeneron Pharmaceuticals
- Sanofi
- Novartis AG
- Amgen Inc.
- Gilead Sciences (Incl. Kite Pharma)
- Eli Lilly and Company
- BeiGene Ltd
- Innovent Biologics
- MacroGenics
- Zymeworks Inc.
- Genmab A/S
- Seagen Inc.
- I-Mab Biopharma
- Hengrui Medicine
- Alphamab Oncology
Segments Covered in the Report
By Drug Class
- Checkpoint Inhibitors
- PD-1 Inhibitors
- PD-L1 Inhibitors
- CTLA-4 Inhibitors
- LAG-3 Inhibitors
- TIGIT Inhibitors
- Others
- Bispecific Antibodies
- T-cell Engagers (BiTEs)
- Dual Immune Checkpoint Blockers
- Dual Tumor Targeting
- Others
- Immune Agonist Antibodies
- OX40 Agonists
- CD137 (4-1BB) Agonists
- Others
- Antibody-Drug Conjugates (ADCs)
- Targeted Payload Carriers
- Immune-Stimulating ADCs
- Others
- Tumor-targeted mAbs with ADCC/CDC
- Radiolabeled Antibodies
- Fusion Protein-Antibody Hybrids
By Application (Cancer Type)
- Non-Small Cell Lung Cancer (NSCLC)
- Melanoma
- Renal Cell Carcinoma
- Head and Neck Cancer
- Triple-Negative Breast Cancer (TNBC)
- Colorectal Cancer (CRC)
- Hepatocellular Carcinoma (HCC)
- Hematologic Malignancies (Lymphoma, Leukemia, Myeloma)
- Bladder Cancer
- Gastric and Esophageal Cancer
- Cervical and Endometrial Cancer
- Others (Pancreatic, Brain Tumors, Rare Cancers)
By Mechanism of Action
- Immune Checkpoint Blockade
- Immune Cell Redirection
- Tumor Antigen Targeting with Immune Effector Recruitment
- Cytokine Pathway Modulation
- Immune Stimulation via Receptor Agonism
- Combination of Immune Activation and Cytotoxicity
By Route of Administration
- Intravenous (IV)
- Subcutaneous (SC)
- Others (e.g., Intratumoral, Intraperitoneal)
- End User
- Hospitals
- Cancer Specialty Clinics
- Academic and Research Institutions
- Others (Outpatient Centers, Homecare)
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Specialty Pharmacies
- Others (Clinical Trial Supply, Direct-to-Patient)
By Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
Get Sample@ https://www.precedenceresearch.com/sample/6513
