The global peptide therapeutics CDMO market expands as pharma and biotech outsource complex peptide drug development and scalable manufacturing
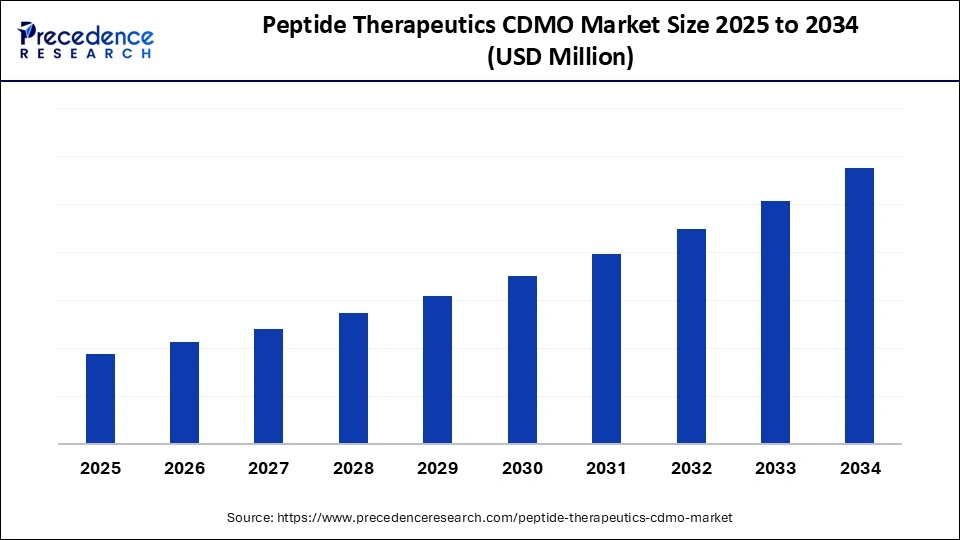
Market Overview
The Peptide Therapeutics CDMO Market has emerged as a cornerstone of modern drug development and manufacturing. Contract Development and Manufacturing Organizations (CDMOs) specializing in peptide therapeutics provide critical services such as synthesis, formulation, scaling, and regulatory compliance. As peptides become increasingly relevant in treating metabolic disorders, cancers, infectious diseases, and rare conditions, demand for outsourced expertise continues to rise.
Peptide-based drugs occupy a unique niche between small molecules and biologics, offering targeted mechanisms of action with fewer side effects. Their complexity, however, requires advanced infrastructure and specialized knowledge, which is why pharmaceutical companies increasingly rely on CDMOs. The growth trajectory of the Peptide Therapeutics CDMO Market reflects rising healthcare expenditures, increasing R&D collaborations, and favorable regulatory frameworks that support peptide drug approvals.
AI and Innovation
Artificial intelligence is transforming the Peptide Therapeutics CDMO Market by accelerating every stage of the drug development lifecycle. AI algorithms now assist in peptide sequence design, predicting stability, bioavailability, and binding affinity. This drastically reduces trial-and-error approaches in early-stage research.
In manufacturing, AI-powered predictive maintenance ensures equipment efficiency and minimizes downtime. Robotics and automation further enhance the precision of peptide synthesis and purification processes. Meanwhile, digital twins of manufacturing systems allow CDMOs to simulate production scenarios, ensuring scalability before committing to large-scale runs.
These innovations make CDMOs not just service providers, but strategic partners in advancing the next generation of peptide therapeutics. As AI becomes integral to discovery and development, the Peptide Therapeutics CDMO Market is well-positioned to lead innovation in the pharmaceutical industry.
Future Trends of Market
The future of the Peptide Therapeutics CDMO Market is defined by several converging trends. First, the rise of personalized medicine is boosting demand for custom peptide synthesis tailored to individual genetic and clinical profiles. CDMOs with capabilities in small-batch, flexible manufacturing are gaining an advantage.
Second, regulatory agencies are streamlining approval pathways for peptide-based drugs, encouraging more biotech startups to enter the space. This creates fresh opportunities for CDMOs to partner with smaller firms that lack in-house expertise.
Third, the adoption of continuous manufacturing processes will improve efficiency and reduce costs, making peptide drugs more accessible. With advances in green chemistry and sustainable practices, the Peptide Therapeutics CDMO Market will also align with global environmental goals.
Rising Demands of Market
The Peptide Therapeutics CDMO Market is experiencing rising demand fueled by both clinical and commercial forces. On the clinical side, peptides are increasingly being investigated as therapies for chronic diseases such as diabetes, obesity, and cardiovascular conditions. Their targeted mechanism of action and lower toxicity make them ideal candidates for long-term treatment regimens.
On the commercial side, pharmaceutical companies are under pressure to reduce costs and speed up time-to-market. Outsourcing to CDMOs allows them to focus on discovery while leveraging external expertise for manufacturing and regulatory compliance. The rising prevalence of rare diseases and orphan drug designations further accelerates demand, as smaller patient populations require specialized and efficient production methods.
Key Market Highlights
Several key highlights define the Peptide Therapeutics CDMO Market:
-
Growing Outsourcing Demand – Both large pharma and biotech startups are increasingly outsourcing peptide production to reduce costs.
-
Regulatory Support – Orphan drug incentives and fast-track approvals are boosting peptide drug pipelines.
-
R&D Collaborations – Partnerships between CDMOs and research institutes are expanding innovation.
-
Technological Integration – AI, automation, and continuous processing are redefining production.
-
Global Expansion – CDMOs are setting up facilities across emerging markets to meet rising demand.
Market Growth Drivers
The Peptide Therapeutics CDMO Market is being propelled by several growth drivers. Increasing prevalence of chronic diseases worldwide has created a strong demand for peptide drugs, particularly in metabolic and oncology segments. Peptide therapeutics are also being explored for vaccine development, a trend accelerated by global health challenges.
Another growth driver is the pharmaceutical industry’s shift toward outsourcing. Maintaining peptide synthesis infrastructure in-house is costly, prompting both established companies and startups to rely on CDMOs. Finally, government initiatives promoting rare disease research and biotech innovation contribute to the market’s upward trajectory.
Restraints
Despite its growth, the Peptide Therapeutics CDMO Market faces certain restraints. High production costs remain a significant challenge due to the complex nature of peptide synthesis and purification. Additionally, scalability issues often arise when moving from laboratory-scale to commercial-scale production.
Regulatory hurdles also pose challenges, as stringent quality requirements increase development timelines and costs. Intellectual property concerns can complicate collaborations between CDMOs and biotech firms. Lastly, a shortage of skilled professionals with expertise in peptide chemistry limits the pace of industry expansion.
Opportunities
Opportunities abound in the Peptide Therapeutics CDMO Market. One major opportunity lies in developing advanced delivery systems to improve peptide stability and patient compliance. CDMOs that can integrate formulation expertise with manufacturing will stand out.
Another opportunity is the expansion of services tailored to emerging biotech startups. By offering end-to-end solutions, from preclinical synthesis to commercial manufacturing, CDMOs can capture a broader client base. Additionally, expansion into Asia-Pacific markets, where healthcare investment is surging, presents significant growth potential.
Regional Insights
-
North America leads due to advanced infrastructure, high R&D spending, and a concentration of biotech startups.
-
Europe is a major hub with strong regulatory support and collaborative research networks.
-
Asia-Pacific is the fastest-growing market, fueled by rising healthcare investment in China, India, and South Korea.
-
Latin America and Middle East & Africa are emerging regions with increasing pharmaceutical outsourcing opportunities.
Peptide Therapeutics CDMO Market Companies

- Almac Group
- AmbioPharm Inc.
- American Peptide Company
- Auspep Pty Ltd.
- Bachem AG
- BCN Peptides S.A.
- Chinese Peptide Company (CPC)
- CordenPharma International
- CPC Scientific Inc.
- Creative Peptides
- CSBio Co.
- GenScript Biotech Corporation
- Lonza Group Ltd.
- Peptide Institute Inc.
- Piramal Pharma Solutions
- PolyPeptide Group
- Sanofi (Zentiva)
- ScinoPharm Taiwan Ltd.
- Siegfried Holding AG
- Thermo Fisher Scientific Inc.
Also Read@ https://www.dailytechbulletin.com/semiconductor-wafer-transfer-robots-market/
