Deoxycholic Acid Obesity Drugs Market Key Takeaways
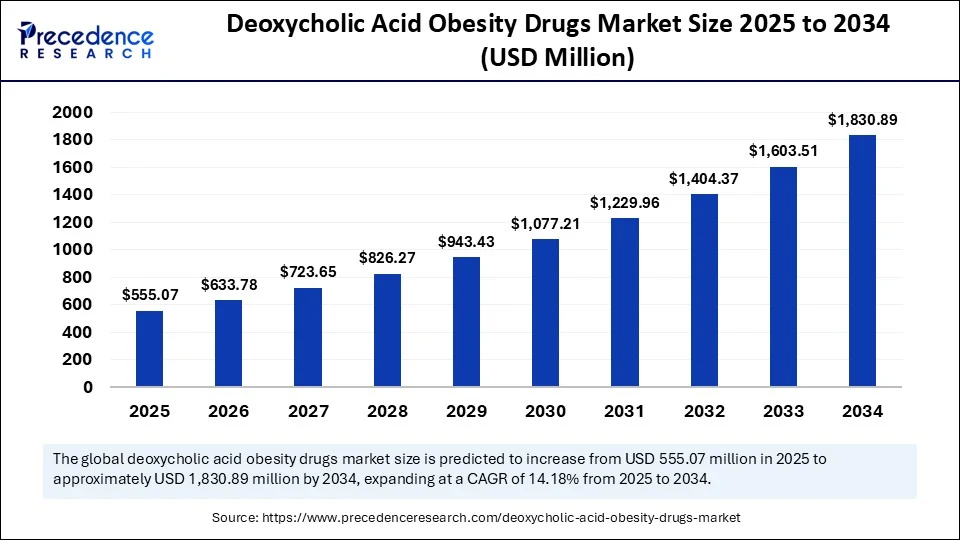
- In terms of revenue, the global deoxycholic acid obesity drugs market was valued at USD 486.14 million in 2024.
- It is projected to reach USD 1,830.89 million by 2034.
- The market is expected to grow at a CAGR of 14.18% from 2025 to 2034.
- North America led the global deoxycholic acid obesity drugs market with the largest market share of 67% in 2024.
- Asia Pacific is estimated to expand at the fastest CAGR between 2025 and 2034.
- By drug type, the ATX-101 (Deoxycholic Acid Injection / Kybella / Belkyra) segment held the biggest market share of 94% in 2024.
- By drug type, the experimental / compounded deoxycholic acid formulations segment is anticipated to grow at a remarkable CAGR between 2025 and 2034.
- By application, the submental fat reduction (double chin) segment captured the highest 88% of market share in 2024.
- By application, the abdominal fat reduction (off-label) segment is expected to expand at a notable CAGR over the projected period.
- By end user, the aesthetic clinics & medspas segment contributed the major market share of 61% in 2024.
- By end user, the plastic surgery centers segment is anticipated to grow at a remarkable CAGR between 2025 and 2034.
- By distribution channel, the physician-dispensed / in-clinic administration segment generated the largest market share of 84% share in 2024.
- By patient demographic, the adults aged 25–45 years segment accounted for the significant market share of 38.1% in 2024.
- By patient demographic, the 46–60 Years segment is expected to expand at a notable CAGR over the projected period.
AI Advances Development of Deoxycholic Acid-Driven Obesity Drugs
Artificial intelligence is emerging as a powerful tool in the development of novel deoxycholic acid (DCA)-based therapies for obesity. AI is playing an increasingly critical role in advancing the DCA obesity drug market, particularly in the design of peptide-based compounds.
A 2025 patent outlines a DCA-peptide product engineered to inhibit adipogenesis and boost lipolysis, signaling a more targeted strategy for obesity management.
Recent research also highlights DCA’s ability to enhance insulin sensitivity and stimulate thermogenesis by promoting fat breakdown and improving mitochondrial function in animal models. These newly uncovered mechanisms present opportunities for AI-driven molecular modeling to further optimize therapeutic candidates. Taken together, these developments position AI as a key enabler in accelerating the discovery and refinement of next-generation DCA-based obesity treatments.
Market Overview
The market for deoxycholic acid obesity drugs market is at the intersection of pharmacological innovation and non‑surgical cosmetic intervention. Deoxycholic acid, a bile acid that disrupts fat cell membranes, has achieved regulatory approval for localized fat reduction (e.g. submental fullness) and is now being evaluated in broader aesthetic and obesity management contexts.
With increasing consumer interest in minimally invasive weight and fat‑loss therapies, pharmaceutical and aesthetic device companies are investing in formulation improvements and clinical trials to expand indications.
The market is emerging robustly in med‑spa and dermatology settings, complemented by clinical research exploring its use in metabolic obesity management. Growth forecasts project steady expansion as formulation enhancements—injectable, encapsulated, or systemic—seek FDA and global approvals.
Drivers
One key driver is consumer demand for non‑surgical fat reduction with minimal downtime. Injectables that dissolve fat without liposuction attract both cosmetic and medical clientele. Positive clinical safety and efficacy data from existing deoxycholate injections boost provider confidence and patient acceptance.
Regulatory milestone achievements—initial approvals for submental fat—pave the way for label expansion and higher insurance interest. Innovation in delivery technology (e.g. controlled formulations, micro‑encapsulation, combination with lipase inhibitors) enhances pharmacokinetics and efficacy. Finally, aesthetic industry expansion and med‑spa proliferation mobilizes broader adoption, especially in major urban centers.
Opportunities
The biggest opportunity lies in seeking broader obesity‑management indications, such as small localized fat pockets in abdomen or thighs. Pharmaceutical companies can develop controlled‑release or targeted systemic deoxycholic acid formulations to treat subcutaneous or visceral fat minimally invasively. Clinical combination therapies—pairing deoxycholate with GLP‑1 analogues or lipase inhibitors—may offer synergistic fat‑reduction pathways.
Emerging markets expansion in Asia, Latin America, and the Middle East offers potential for cosmetic adoption. Research‑driven proof of metabolic benefit (e.g. improvements in insulin sensitivity or lipid profile) could pivot the drug toward metabolic obesity treatment rather than purely cosmetic. Finally, platform differentiation through nano‑formulations or localized injection systems offers competitive advantage.
Challenges
However, the market faces substantial hurdles. Safety concerns around injection‑site reactions, nerve injury, and variable fat reduction require careful dosage optimization and training. Limited indication scope — currently approved for submental fat only — restricts market size unless label expansion is achieved. Injection discomfort and multiple‑session necessity may deter patients compared to oral obesity medication.
Competition from systemic obesity drugs (e.g. GLP‑1 receptor agonists) overshadow deoxycholic acid’s cosmetic positioning. Regulatory complexity in approving systemic or broader-use formulations is high; long duration safety and metabolic trials are needed. Pricing and reimbursement challenge: unless recognized for metabolic indication, payers typically classify it as elective cosmetic treatment, limiting coverage.
Recent Developments
In recent clinical pipelines, a mid‑stage trial investigating deoxycholic acid micro‑injections for thigh and bra‑line fat reduction demonstrated favorable safety and dose‑dependent efficacy. A biotech firm partnered with a pharmaceutical house to explore systemic encapsulated deoxycholic acid for localized visceral fat targeting.
Another company launched a reformulated deoxycholate injection with longer persistence and fewer injections needed per session. Dermal clinics expanded off-label use under physician supervision, driving informal demand. Preclinical studies showed deoxycholic acid combined with adipocyte‑modulating hormones improves metabolic markers.
Clinical conferences spotlighted emerging data on combination protocols with injectable obesity drugs for synergistic fat loss. Regulatory agencies in several countries have issued draft guidance for off‑label use and physician‑administered protocols. New training academies for aesthetic injectors have started offering certification programs specific to deoxycholate administration, aiming to reduce risks and standardize protocols.
Deoxycholic Acid Obesity Drugs Market Companies
- AbbVie Inc.
- Revance Therapeutics, Inc.
- Ipsen
- Hugel, Inc. (South Korea)
- Medytox, Inc.
- Daewoong Pharmaceutical Co., Ltd.
- Endo International plc
- Venus Remedies
- Allergan Aesthetics (subsidiary of AbbVie)
- Revexia Life Sciences
- Contura International Ltd.
- OBE Pharma
- NovaBay Pharmaceuticals
- Laboratoires VIVACY (France)
- Sinclair Pharma
- Hyundae Meditech (Korea)
- DermaSculpt
- SinoPharm Group
- Croma Pharma
- Anika Therapeutics
Segments Covered in the Report
By Drug Type
- ATX-101 (Deoxycholic Acid Injection / Kybella / Belkyra)
- Experimental / Compounded Deoxycholic Acid Formulations
- Combination Therapies (under development)
- Oral Deoxycholic Acid Products (limited and non-FDA approved)
By Application
- Submental Fat Reduction (Double Chin)
- Abdominal Fat Reduction (off-label)
- Thighs, Arms, and Flanks (off-label aesthetic uses)
- Lipomas and Localized Fat Deposits
- Cellulite Management (experimental)
By End User
- Aesthetic Clinics & MedSpas
- Dermatology Clinics
- Plastic Surgery Centers
- Hospitals
- Home Administration (emerging, under supervision)
By Distribution Channel
- Physician-Dispensed / In-Clinic Administration
- Retail Pharmacies (in regions where approved for direct sales)
- Online Pharmacies (regulated channels only)
By Patient Demographics
- Adults Aged 25–45 Years
- 46–60 Years
- Millennials Seeking Non-Surgical Aesthetic Procedures
- Men
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Get Free Sample @ https://www.precedenceresearch.com/sample/6502
