A Market Evolving with Science and Strategy
The Contract Development and Manufacturing Organizations (CDMOs) market is entering a transformative decade between 2025 and 2034. What was once seen as a cost-cutting measure has become a strategic growth enabler for pharmaceutical and biotech companies. The rise of complex biologics, the personalization of therapies, and rapid technological integration have driven a strong shift from traditional outsourcing to long-term CDMO partnerships. Today’s CDMOs are no longer just suppliers—they are strategic collaborators supporting innovation, speed, and global scalability.
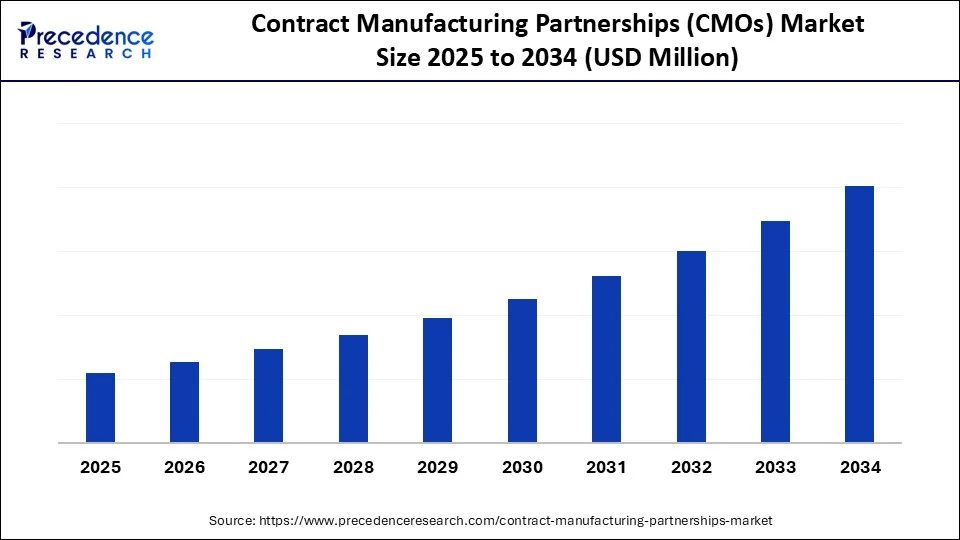
Biologics, Personalized Therapies, and the Rise of End-to-End Solutions
One of the most significant growth drivers in the CDMO landscape is the surging demand for biologics—monoclonal antibodies, cell and gene therapies, and next-generation vaccines. Biopharma companies are increasingly leaning on CDMOs for not only manufacturing but also for upstream process development, analytical testing, formulation, fill-finish, and regulatory documentation. This shift has propelled CDMOs toward offering integrated, end-to-end services.
Simultaneously, the growing emphasis on personalized medicine has reshaped manufacturing timelines and batch sizes. With customized therapies on the rise, pharma companies require agile, high-tech partners capable of adaptive manufacturing strategies, often across global geographies. This evolution has redefined how outsourcing is perceived—not just as a tactical move, but a foundational strategy.
Get a Sample: https://www.precedenceresearch.com/sample/6434
Strategic Alliances and Capacity Partnerships
A notable trend reshaping the CDMO space is the rise of long-term strategic alliances and dedicated capacity deals. Biotech firms, in particular, are securing exclusive lines or facilities within CDMOs to lock in manufacturing security. This model reduces supply chain risks, ensures regulatory alignment, and speeds up market access. For CDMOs, such partnerships offer a predictable revenue stream and a pathway to co-develop innovative technologies or therapies.
Examples of such collaborations include the Thermo Fisher-Sanofi alliance for biologics and Zydus’ tie-up with Agenus for immuno-oncology solutions. These deals indicate a shift away from transactional outsourcing to deeply integrated, innovation-driven alliances.
AI, Automation, and the Future of Smart Manufacturing
Technology is the backbone of the next-generation CDMO. Artificial Intelligence is being rapidly integrated into various facets—from real-time quality assurance using AI-driven vision systems to adaptive process controls powered by machine learning. Predictive analytics is streamlining maintenance schedules, while digital twins are helping simulate and optimize manufacturing processes in silico before physical production even begins.
Automation and robotics are also redefining operations—minimizing human error, enhancing sterility in biologics manufacturing, and reducing turnaround time. CDMOs are increasingly investing in modular and continuous manufacturing systems to support just-in-time delivery models. Additionally, cloud-based batch tracking and the use of blockchain are becoming critical tools for auditability, traceability, and regulatory compliance in global markets.
Market Scope
| Report Coverage | Details |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Service Type, Drug Type, Business Model, Scale of Operation, End User, Therapeutic Area, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Trends: Small Molecule, Biologics, and Cell & Gene Therapy
The market remains split between small and large molecule manufacturing, but biologics are clearly taking the lead in terms of growth rate. CDMOs are racing to expand bioreactor capacity, set up single-use technologies, and comply with GMP requirements for high-potency products.
Meanwhile, cell and gene therapy CDMOs are emerging as a specialized niche. These facilities must meet exceptionally high standards in terms of sterility, scalability, and patient-specific manufacturing. As the FDA and EMA continue approving more advanced therapies, the demand for CDMOs equipped for viral vector production and autologous cell processing is poised to rise sharply.
Fee-for-service remains a popular engagement model, but a growing number of players are experimenting with risk-sharing and milestone-based models, especially in the development phase.
Challenges: Capacity, Compliance, and Control
Despite the growth, the CDMO sector is not without hurdles. Capacity constraints, especially in biologics, are tightening lead times and inflating costs. As regulatory bodies demand more stringent GMP compliance, CDMOs must invest heavily in quality infrastructure and documentation systems.
Vendor reliability and data/IP security are also top concerns. With sensitive clinical and commercial data being exchanged, companies are increasingly scrutinizing CDMO partners for cybersecurity readiness and confidentiality protocols.
Global Growth Hotspots: Asia-Pacific, LATAM, and the Nearshoring Wave
While the U.S. and Europe remain the epicenters of high-quality GMP manufacturing, emerging markets are making bold moves. Asia-Pacific, led by India and China, is gaining ground through cost-efficient yet increasingly sophisticated CDMO offerings. Both countries are investing in biologics capacity and high-potency active pharmaceutical ingredient (HPAPI) infrastructure.
Latin America, especially Brazil and Mexico, is witnessing a surge in demand for localized manufacturing as regional health policies emphasize drug affordability and self-reliance. Meanwhile, in North America, reshoring and nearshoring strategies are gaining momentum. To de-risk supply chains, U.S.-based pharma firms are looking to establish or partner with CDMOs closer to home.
Read Also: Biopharma Clinical Trials Market Size, Share, Report by 2034
Opportunities on the Horizon
Over the next decade, CDMOs specializing in niche high-value products—such as oligonucleotides, lipid nanoparticles, and viral vectors—will find themselves in high demand. Capacity expansion in emerging economies, paired with digital innovation, is creating a fertile landscape for startups and virtual biotech companies that require development and manufacturing support without massive in-house investment.
Moreover, sustainability is becoming a strategic priority. CDMOs that integrate green chemistry, energy-efficient processes, and ESG-compliant practices will be more attractive to pharma firms under pressure to meet environmental goals.
Competitive Landscape: Leaders, M&A, and Innovation Hubs
The CDMO market is witnessing aggressive consolidation, with major players expanding capacity through acquisitions and partnerships. Lonza, Thermo Fisher’s Patheon, Catalent, Samsung Biologics, and WuXi Biologics continue to dominate due to their broad service portfolios and global infrastructure.
Innovation partnerships are also thriving. From AI software firms collaborating with manufacturing units to universities co-developing analytical platforms, the CDMO ecosystem is becoming increasingly collaborative and tech-driven. M&A activities are geared toward bolstering biologics capabilities and entering new geographic markets.
