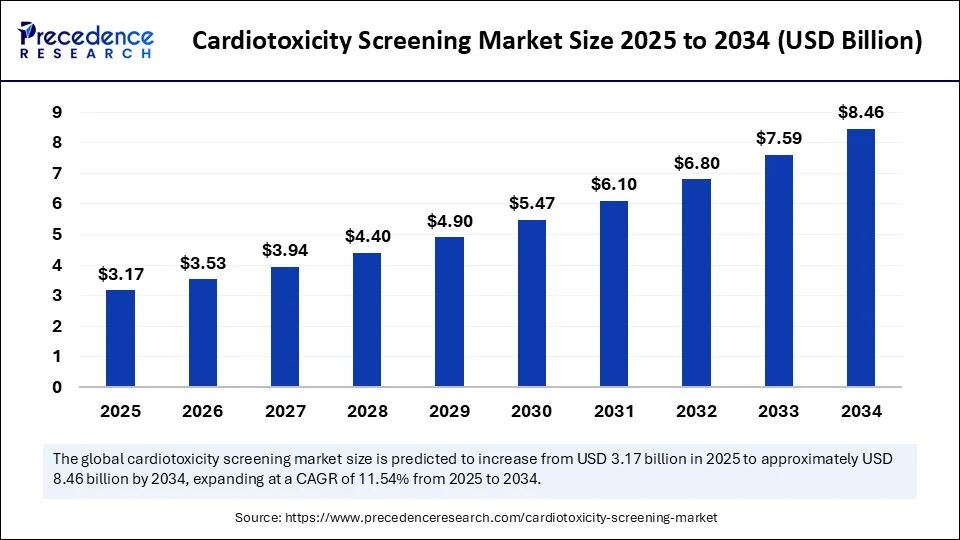
Market Overview
The cardiotoxicity screening market has gained significant traction in recent years as cardiovascular safety has become a crucial concern in drug development and healthcare diagnostics. Cardiotoxicity, which refers to damage to the heart muscle or its function due to chemical substances, particularly pharmaceuticals, necessitates robust screening technologies to identify and mitigate risks early. This market encompasses a broad range of technologies, including in vitro assays, imaging techniques, and biomarker testing that help detect potential cardiac toxicity. Growing awareness about drug-induced cardiotoxicity and stringent regulatory requirements are driving the expansion of the cardiotoxicity screening market globally.
AI and Innovation
Artificial intelligence and innovative technologies are playing transformative roles in the cardiotoxicity screening market. AI-powered algorithms enable rapid data processing and predictive modeling to assess the cardiotoxic potential of new chemical entities with higher accuracy and efficiency. Machine learning techniques analyze vast datasets derived from high-throughput screening and clinical trials to identify subtle cardiac risk signals that might be overlooked by traditional methods.
Innovations in organ-on-chip models and 3D bioprinting are improving the physiological relevance of cardiotoxicity assays. These advances collectively boost the sensitivity and specificity of cardiotoxicity screening, positioning the market for accelerated growth.
Future Trends of Market
The future of the cardiotoxicity screening market is shaped by several emerging trends, including the integration of multi-omics technologies such as genomics, proteomics, and metabolomics to provide comprehensive cardiac safety profiles. There is increasing adoption of patient-specific induced pluripotent stem cell (iPSC)-derived cardiomyocytes, enabling personalized cardiotoxicity assessments.
Additionally, the use of digital biomarkers and wearable devices to monitor cardiac function in real time is expected to expand. Regulatory agencies are progressively encouraging the use of advanced in vitro and in silico models, fostering innovation. These trends are likely to enhance predictive accuracy and reduce drug attrition rates, driving the cardiotoxicity screening market forward.
Rising Demands of Market
Rising demand in the cardiotoxicity screening market is driven by the expanding pharmaceutical pipeline and the increasing complexity of new drug candidates, which often possess potential cardiac risks. Regulatory bodies are imposing stricter requirements for preclinical and clinical cardiac safety evaluations, fueling the need for advanced screening solutions.
Furthermore, the growing prevalence of cardiovascular diseases worldwide necessitates better safety profiling of medications to prevent adverse cardiac events. Increased R&D spending by pharmaceutical companies and growing awareness among healthcare professionals regarding cardiotoxic risks contribute significantly to the market’s rising demand.
Key Market Highlights
Key highlights of the cardiotoxicity screening market include the advent of high-throughput screening technologies that enable rapid assessment of multiple compounds. Strategic partnerships between biotechnology firms and pharmaceutical companies aim to co-develop enhanced screening platforms. The launch of novel cardiotoxicity biomarkers and non-invasive imaging technologies has broadened the toolkit available for cardiac safety evaluation. Regulatory guidelines emphasizing early detection and risk mitigation of cardiotoxicity underscore the critical role of this market. Moreover, investments in research focused on reducing drug development costs by minimizing late-stage failures highlight the market’s importance.
Market Growth Drivers
Several drivers propel the cardiotoxicity screening market, including the rising number of drug candidates with potential cardiotoxic effects, necessitating comprehensive safety testing. Increasing regulatory scrutiny and the enforcement of stringent cardiac safety guidelines drive pharmaceutical companies to adopt advanced screening tools. The rising incidence of cardiovascular diseases globally elevates the need for safer drugs, indirectly stimulating market growth.
Technological advancements such as high-content screening and the development of human-relevant models contribute to improved detection capabilities. Growing collaborations between academia and industry further support market expansion.
Restraints
Despite its growth potential, the cardiotoxicity screening market faces certain restraints. High costs associated with advanced screening technologies may limit adoption, particularly among smaller biotech firms and in emerging economies. The complexity of accurately modeling human cardiac physiology in vitro poses scientific challenges that can affect screening reliability. Additionally, regulatory approval processes for novel screening methods can be lengthy and uncertain. Variability in biomarker expression and interpretation may complicate data analysis. These factors may hinder the rapid scaling of cardiotoxicity screening solutions.
Opportunities
The cardiotoxicity screening market presents numerous opportunities, particularly in emerging markets with growing pharmaceutical R&D activities. Development of automated, high-throughput screening platforms offers scalability and cost reduction potential. The integration of AI-driven predictive models can revolutionize early-stage drug safety assessments. Opportunities also lie in expanding the application of cardiotoxicity screening beyond pharmaceuticals, such as in environmental toxicology and chemical safety. Collaboration opportunities between technology providers, pharmaceutical companies, and regulatory bodies can foster innovation and standardization, further strengthening the market.
Also Read@ https://www.dailytechbulletin.com/semiconductor-wafer-transfer-robots-market/
Cardiotoxicity Screening Market Companies

- Charles River Laboratories
- Labcorp/Covance
- Eurofins Scientific
- WuXi AppTec
- Evotec
- ICON plc
- Certara
- Simulations Plus
- Axion BioSystems
- Multi-Channel Systems (MCS)
- Nanion Technologies
- Sophion Bioscience
- Molecular Devices/Danaher group
- Fujifilm-CDI (Cellular Dynamics International)
- Ncardia (formerly Axiogenesis)
- Axol Bioscience
- Thermo Fisher Scientific
- PerkinElmer
- Charles River/HESI/CiPA collaborators
Recent Developments
- In June 2025, the U.S. Geological Survey published in the journal Environmental Science and Technology, identifying Biosphenol P (BPP) as a potent cardiotoxic compound through high-throughput screening. Researchers have highlighted BPP’s ability to induce cardiotoxicity by apoptosis and the NF-kB pathway.
- In April 2025, the industry-leading solutions to streamline high-throughput screening while maintaining physiological relevance were launched by the Ncardia in a “Advancing high-throughput cardiac screening with physiologically relevant 3D iPSC-based models,” focused webinar.
Get Free sample link @ https://www.precedenceresearch.com/sample/6624
