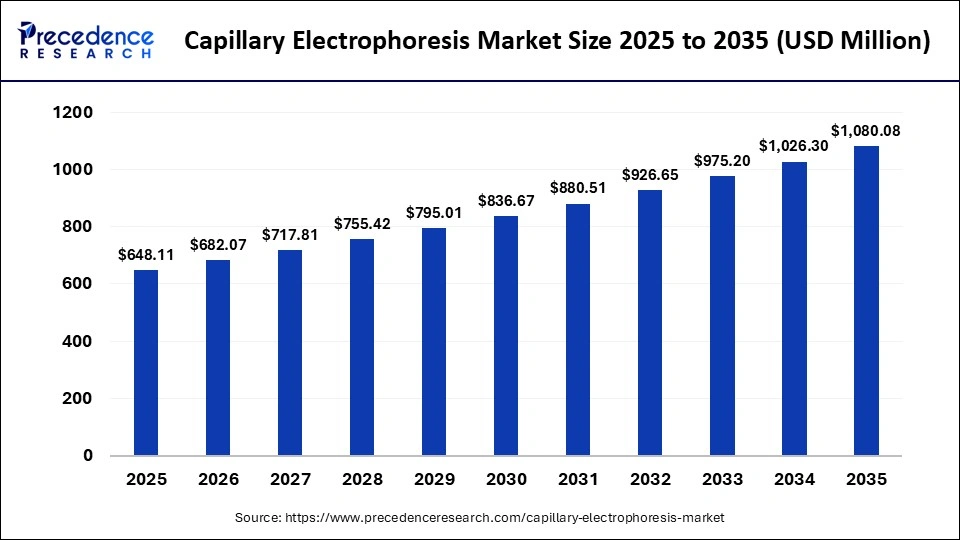
Market Key Takeaways
- North America dominated the market with the highest market share of 46.8% in 2025.
- The Asia Pacific is expected to grow at a notable CAGR 7% between 2026 and 2035.
- By product type, the capillary electrophoresis systems segment contributed the biggest market share of 58.8% in 2025.
- By product type, the reagents & consumables segment growing at a solid CAGR of 6.2% between 2026 and 2035.
- By technique, the capillary zone electrophoresis (CZE) segment captured the highest market share of 42.5% in 2025.
- By technique, the capillary isoelectric focusing (CIEF) segment is set to grow at a 6.1% CAGR between 2026 and 2035.
- By end-user, the pharmaceutical & biotechnology companies segment registered dominance by capturing 35.5% of the market share in 2025.
- By end-user, the clinical laboratories segment is poised to grow at a 5.9% of CAGR from 2025 to 2034.
- By application, the drug development and QC segment held a dominant market share of 38.9% in 2025.
- By application, the clinical diagnostics segment is growing at a CAGR of 5.8% between 2026 and 2035.
Market Overview
The capillary electrophoresis (CE) market is witnessing robust growth, driven by increasing demand for high-resolution analytical techniques in pharmaceutical research, drug development, and quality control. CE is an electrokinetic separation method that uses an electric field to separate charged analytes within narrow fused-silica capillaries based on their charge-to-size ratio. The technique requires minimal sample volumes, generates low waste, and delivers highly reproducible results with sharp, well-defined peaks, making it well suited for modern analytical laboratories.
Adoption of CE is expanding as pharmaceutical and biotechnology companies intensify efforts to characterise complex biologics, detect trace impurities, assess glycosylation patterns, and identify charge variants processes that are critical for ensuring product safety and meeting stringent regulatory requirements. Technological advancements, particularly the integration of capillary electrophoresis with mass spectrometry (CE-MS), have significantly broadened analytical capabilities and sensitivity. Beyond drug development, CE is increasingly used in clinical laboratories and research institutions, and rising global demand for biologics and advanced diagnostics is expected to further support market growth.
Impact of Artificial Intelligence on the Capillary Electrophoresis Market
Artificial intelligence is playing an increasingly important role in advancing the capillary electrophoresis market by enhancing innovation, improving analytical efficiency, and increasing the overall value of CE systems. AI-powered data analysis tools enable laboratories to interpret electropherograms with greater accuracy and speed, reducing manual intervention and analytical errors. Instrument developers are embedding AI into CE platforms to support automated method development, predict separation performance, and dynamically optimize operating parameters.
As AI adoption grows, CE is finding expanded use in high-value applications such as therapeutic development, clinical diagnostics, environmental monitoring, and food safety testing. These AI-enabled capabilities are helping laboratories achieve higher throughput, better reproducibility, and more insightful data interpretation, positioning capillary electrophoresis as a smarter, more versatile analytical technology.
Capillary Electrophoresis Market Growth Factors
- Rising Biopharmaceutical R&D Activities: Increasing development of biologics and biosimilars is driving strong demand for high-resolution capillary electrophoresis techniques to support detailed molecular characterization and quality assessment.
- Expanding Clinical Diagnostic Applications: Growing use of CE in protein profiling, genetic testing, and biomarker analysis is contributing to broader adoption in clinical and research laboratories.
- Growing Adoption of Automated CE Systems: Automation in sample preparation, separation, and data analysis is improving laboratory efficiency, reducing manual errors, and increasing analytical throughput.
- Advancements in CE–MS Integration: Enhanced coupling of capillary electrophoresis with mass spectrometry is expanding analytical capabilities, enabling deeper molecular insights and more precise characterization.
- Increasing Regulatory Compliance Requirements: Stricter regulatory standards for drug quality control, impurity profiling, and biopharmaceutical validation are accelerating the adoption of CE across pharmaceutical and biotechnology industries.
Regional Insights: Capillary Electrophoresis Market
North America holds a leading position in the capillary electrophoresis market, driven by strong pharmaceutical and biotechnology industries, advanced laboratory infrastructure, and high adoption of innovative analytical technologies. The United States, in particular, benefits from extensive biologics R&D, stringent regulatory requirements, and early integration of AI-enabled CE systems in drug development and quality control.
Europe represents a mature and steadily growing market, supported by a strong focus on biologics manufacturing, regulatory compliance, and academic research. Countries such as Germany, the UK, France, and Switzerland are key contributors, with widespread use of CE in pharmaceutical analysis, clinical research, and food safety testing. Increasing investment in CE–MS platforms and automation is further strengthening market growth.
The Asia Pacific region is the fastest-growing market, fueled by rapid expansion of pharmaceutical manufacturing, increasing biologics production, and rising investments in healthcare and research infrastructure. China, Japan, India, and South Korea are major growth drivers, with growing adoption of CE technologies in drug development, diagnostics, and academic research.
In Latin America, the CE market is developing gradually, supported by expanding pharmaceutical industries and growing demand for advanced analytical techniques in quality control and clinical testing. Brazil and Mexico are leading regional adoption as laboratory capabilities continue to improve.
The Middle East & Africa region is at an early stage of adoption, with growth driven by increasing investments in healthcare infrastructure, clinical laboratories, and academic research. While current penetration remains limited, rising focus on advanced diagnostics and pharmaceutical quality testing is expected to create new opportunities for capillary electrophoresis systems in the coming years.
Recent Developments
- In March 2025, Thermo Fisher Scientific launched the IVDR-compliant SeqStudio Flex Dx Genetic Analyzer. The CE system supports both clinical laboratories and research applications, enabling Sanger sequencing and fragment analysis. It is designed to improve efficiency and provide flexible run schedules. The platform ensures compliance with regulatory mandates while enhancing laboratory workflow.
- In January 2024, Agilent Technologies introduced the automated ProteoAnalyzer CE system for protein analysis. The system offers rapid, high-resolution separation of complex protein mixtures, meeting the needs of pharma, biotech, food analysis, and academic research. Growing interest in monoclonal antibodies and protein therapeutics is driving their adoption. The platform simplifies analytical workflows and increases throughput for protein characterization.
- In July 2024, Sysmex and Hitachi High-Tech announced a collaboration to develop CE-based genetic testing systems. They aim to create efficient, low-cost platforms for broader clinical use. The partnership focuses on optimized genetic testing for individual diseases. This initiative follows joint research and a feasibility study conducted since 2023.
Top Companies in the Capillary Electrophoresis Market & Their Offerings
- Agilent Technologies (U.S.)
- SCIEX (U.S.)
- Thermo Fisher Scientific (U.S.)
- PerkinElmer (U.S.)
- Shimadzu Corporation (Japan)
- Bio-Rad Laboratories (U.S.)
- Metrohm (Switzerland)
Get Sample Link : https://www.precedenceresearch.com/sample/7221
