Introduction: The Shift Toward Personalized and Curative Therapies
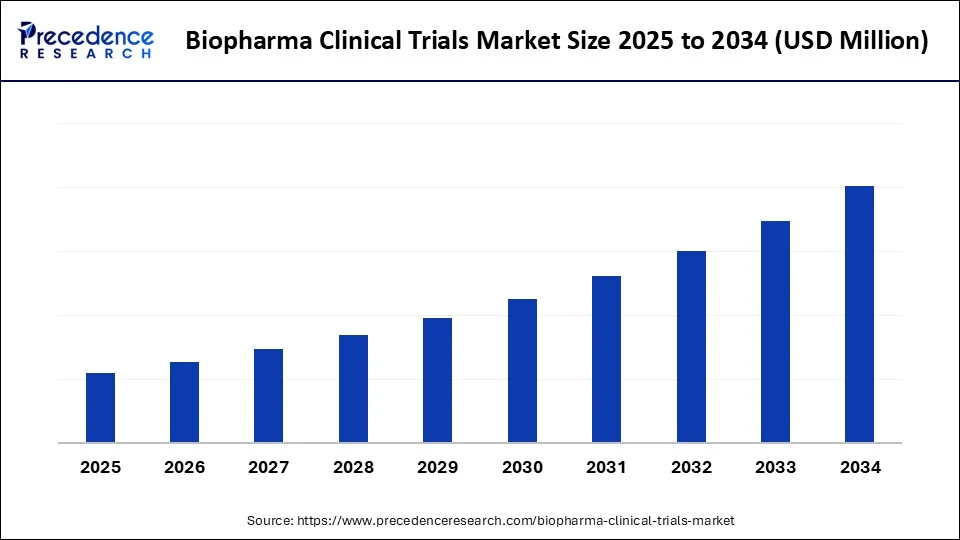
The biopharma clinical trials market is growing with advances in decentralized trials, patient-centric models, and breakthrough therapeutic research.
Cell and gene therapies (CGTs) are redefining the future of medicine. These therapies, which aim to correct the root cause of genetic and chronic illnesses, have emerged as a transformative force in healthcare. Unlike traditional symptom-management treatments, CGTs offer the potential for one-time curative interventions, bringing unprecedented hope to patients with rare or previously untreatable conditions.
This paradigm shift has fueled a rapid increase in the number and complexity of clinical trials tailored to personalized and precision therapies. As a result, the CGT trials market is expanding, with emerging biotech firms and progressive biopharmaceutical companies at the forefront. These innovators are pushing the envelope in research and development, accelerating the availability of next-generation therapies and reshaping the clinical trial landscape.
Market Overview: What Defines the CGT Clinical Trials Ecosystem?
The CGT clinical trials ecosystem is distinct and multifaceted. At its core are investigational therapies such as CAR-T cells, CRISPR gene editing, and other gene-modified platforms. These therapies often require tailored protocols, individualized manufacturing, and sophisticated delivery systems.
This ecosystem is supported by a broad infrastructure that includes clinical trial sponsors, contract research organizations (CROs), contract development and manufacturing organizations (CDMOs), technology vendors, and regulatory authorities. Post-pandemic momentum and a favorable regulatory environment have further bolstered the market. The speed of approvals and increased interest in rare disease therapies are positioning CGT trials as one of the most dynamic segments in clinical research globally.
Get a sample: https://www.precedenceresearch.com/sample/6433
How Are Technology and AI Shaping the Future of CGT Trials?
Technology is revolutionizing how CGT trials are designed, executed, and analyzed. Artificial intelligence and machine learning are now embedded in every phase of the clinical trial process—from identifying eligible patients and optimizing protocols to predicting trial outcomes and reducing delays.
eClinical platforms and decentralized trial models (DCTs) are gaining traction, enabling remote monitoring, digital biomarkers, and enhanced patient engagement. Advanced tools like synthetic control arms and digital twins are reducing reliance on placebo groups and enhancing trial efficiency. Wearables and real-time data collection are further enabling precise monitoring in complex, personalized therapies—bringing a much-needed tech upgrade to the rigorous demands of CGT trials.
Key Market Drivers: What’s Fueling the Growth?
Several powerful forces are driving the surge in CGT clinical trials. Foremost among them is the growing global burden of rare and orphan diseases, many of which lack effective treatments. These unmet medical needs have catalyzed drug developers to invest heavily in CGT platforms.
Supportive regulatory frameworks, such as the FDA’s Breakthrough Therapy and RMAT designations, along with the Orphan Drug Act, are accelerating trial timelines and encouraging innovation. The expanding CGT pipeline—spanning both early-phase and late-stage trials—signals a strong growth trajectory. Additionally, robust investments from pharmaceutical giants and venture capitalists are helping fuel research, infrastructure development, and commercialization strategies.
Challenges Restraining Market Growth
Despite its promise, the CGT trials market is not without challenges. High operational costs, particularly in the manufacturing of autologous therapies, make scalability difficult. Complex logistics involving cold chain storage, time-sensitive deliveries, and personalized dosing add to the financial and technical burden.
Patient recruitment is another significant barrier—especially in rare disease trials where eligible participants are few and geographically dispersed. Regulatory inconsistencies across regions also hinder global trial expansion, as compliance with Good Manufacturing Practices (GMP) and evolving safety standards requires continuous adaptation and oversight.
Opportunities Ahead: Where Is the Market Headed?
The CGT trials market is ripe with opportunity. Collaborations among academic institutions, biotech startups, and CROs are intensifying, creating an environment conducive to innovation and scalability. CDMOs are playing an increasingly vital role by offering turnkey solutions for manufacturing, trial management, and regulatory support.
AI-powered CROs are improving trial timelines through data-driven site selection, real-time analytics, and predictive modeling. Real-world data (RWD) and real-world evidence (RWE) are also gaining prominence, offering powerful insights that can expedite market entry and refine trial design. Technological breakthroughs in gene editing, delivery systems, and non-viral vectors further point to a future rich in both clinical and commercial potential.
Market Segmentation
By Phase
The majority of innovation is currently taking place in early-phase clinical trials (Phase I/II). These stages allow developers to explore novel mechanisms of action and fine-tune gene and cell therapies for safety and efficacy. Adaptive trial designs and basket trials are increasingly utilized to accelerate insights and pivot strategies without restarting the trial process from scratch.
By Therapeutic Area
CGT trials are heavily focused on oncology, hematology, neurology, and rare genetic diseases. Inherited blood disorders like sickle cell disease and hemophilia have been primary targets due to their clear genetic etiology. Spinal muscular atrophy and various cancers—particularly hematologic malignancies—are also at the forefront of CGT research. The therapeutic breadth is expanding, signaling growing interest in metabolic and autoimmune conditions.
By Molecule Type
This segment includes a wide range of therapy types, such as gene-modified cell therapies (e.g., CAR-T, TCR), in vivo and ex vivo gene therapies, and RNA-based therapeutics. Viral vectors (AAV, lentivirus) continue to dominate delivery methods, although there is a growing push toward non-viral and nanoparticle systems that promise improved safety and scalability.
By Sponsor Type
While large pharmaceutical firms are actively investing in CGT trials, much of the disruptive innovation is emerging from smaller biotech companies. Academic institutions and government-funded research organizations are also playing a pivotal role, often partnering with CROs to translate early-stage research into clinical development.
By Study Design
Interventional trials remain the most common format, but hybrid and decentralized trial designs are quickly gaining traction. These models allow for greater flexibility, real-time patient engagement, and the use of digital endpoints. Remote patient monitoring and electronic data capture are now seen as essential tools for improving compliance and trial efficiency.
By End User
The end-user landscape includes biopharmaceutical companies, academic research centers, hospitals, and CROs. Hospitals and specialty clinics are critical for trial execution, particularly in autologous cell therapies, while CROs provide backbone support in trial management and analytics.
By Region
North America continues to lead the global market, supported by strong regulatory frameworks (FDA), robust R&D investment, and a highly developed CRO ecosystem. Europe follows closely, with support from the European Medicines Agency (EMA), advanced healthcare infrastructure, and targeted funding for rare diseases. Asia Pacific is emerging as a key player, especially in countries like China, Japan, and South Korea, where cost-effective trial execution and government support are strong. Latin America and the Middle East & Africa are seeing early-stage developments with increasing interest in building trial infrastructure.
Competitive Landscpe & Key Players
The CGT clinical trials market is populated by both pioneering biotechs and established pharmaceutical companies. Notable players include Novartis, CRISPR Therapeutics, Sangamo Therapeutics, bluebird bio, Spark Therapeutics, Editas Medicine, Precision BioSciences, Autolus Therapeutics, and Verve Therapeutics.
Equally vital are the CROs and CDMOs enabling trial execution and scalability. Leaders like Catalent, Lonza, Charles River Laboratories, and WuXi Advanced Therapies are forming strategic partnerships to support the complex demands of CGT trials.
Recent Developments and Strategic Collaborations
The CGT trials space has seen a surge in strategic activity. From the FDA’s approval of new CAR-T therapies to the first CRISPR-based treatments reaching late-stage trials, regulatory wins are reshaping the market. Mergers and acquisitions are streamlining capabilities, while AI tools are being adopted to optimize protocol design and accelerate patient recruitment. These developments signal a maturing market entering a phase of accelerated execution and delivery.
