Auricular Bioelectronic Devices Market analysis explores growth drivers, challenges, and opportunities in the evolving healthcare technology sector.
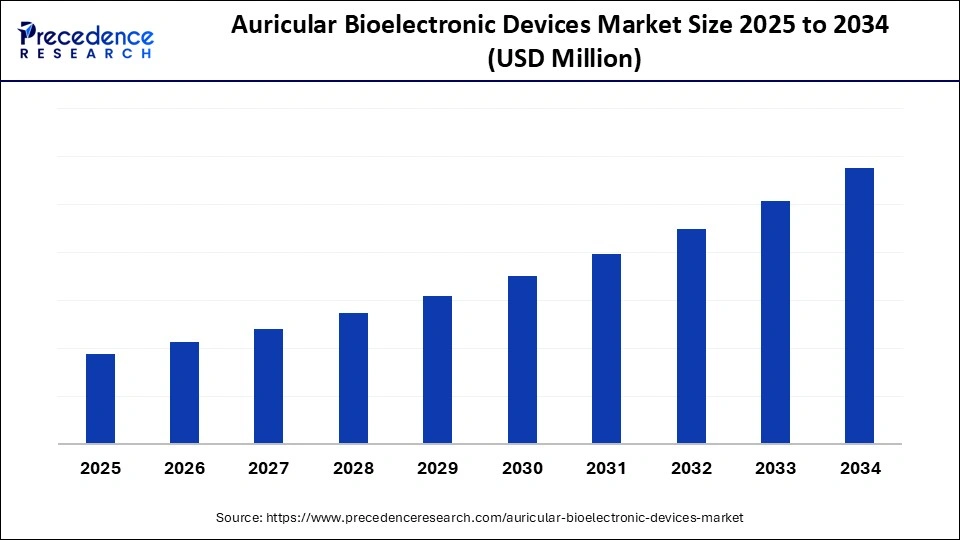
Market Overview
The Auricular Bioelectronic Devices Market is transforming healthcare by introducing non-invasive therapeutic solutions that target neurological and chronic conditions through ear-based electrical stimulation. Unlike traditional pharmaceuticals, these devices rely on the principles of neuromodulation, offering precise and safer alternatives for managing pain, stress, depression, addiction, and sleep disorders.
The market has gained momentum as healthcare providers recognize the value of bioelectronic medicine in reducing dependency on drugs and minimizing side effects. With rising healthcare costs, there is a growing emphasis on cost-effective treatments that improve patient outcomes. This has created favorable conditions for the Auricular Bioelectronic Devices Market to expand, supported by advancements in wearable technologies, sensor miniaturization, and patient-friendly device designs.
AI and Innovation
Artificial intelligence is becoming a cornerstone of the Auricular Bioelectronic Devices Market. Through AI integration, devices can analyze patient signals in real time, adjust stimulation parameters, and provide personalized therapy. Machine learning algorithms further enhance predictive capabilities, allowing doctors to monitor treatment effectiveness and adapt protocols accordingly.
Innovation is also driving significant progress in device design. Lightweight, wireless, and mobile-connected devices are replacing older, bulky models. Integration with smartphone apps enables continuous monitoring and direct communication between patients and healthcare providers. At the same time, material innovations are improving patient comfort, making devices more discreet and durable.
AI and innovation together are accelerating the evolution of auricular bioelectronic devices from niche applications into mainstream healthcare solutions, ensuring better clinical outcomes and higher patient engagement.
Future Trends of Market
Looking ahead, the Auricular Bioelectronic Devices Market is expected to see transformative shifts. Personalized medicine is likely to dominate future developments, as devices are tailored to individual health profiles. For example, stimulation patterns may soon be adapted based on genetic markers, lifestyle patterns, and continuous physiological data.
Another trend shaping the market is the expansion of therapeutic applications. While pain management has been the primary use case, auricular bioelectronic devices are increasingly being studied for treating conditions such as epilepsy, post-traumatic stress disorder, and autoimmune diseases.
Additionally, the convergence of digital health ecosystems with bioelectronic devices will become more prominent. Cloud-based platforms, wearable sensors, and AI-driven analytics will collectively enhance the value proposition of these devices. This interconnected ecosystem will allow patients to receive more comprehensive, data-driven care.
Rising Demands of Market
The demand for devices within the Auricular Bioelectronic Devices Market is climbing rapidly, primarily due to the global surge in lifestyle-related disorders. Stress, chronic pain, and neurological conditions are affecting millions of individuals worldwide, creating an urgent need for effective yet non-invasive therapies.
Patients are also becoming more aware of alternatives to pharmaceutical treatments. Concerns about side effects, drug dependency, and long-term medication costs are pushing individuals to seek safer solutions. The convenience of at-home treatment, coupled with the acceptance of wearable health devices, is further driving demand.
Healthcare systems are also under pressure to deliver efficient care at lower costs. Auricular bioelectronic devices align with this need by providing accessible, scalable, and patient-friendly options.
Key Market Highlights
-
The Auricular Bioelectronic Devices Market is expanding as non-invasive therapies gain wider acceptance.
-
AI and innovation are central to advancing device capabilities and user experience.
-
Growing demand is fueled by rising chronic illnesses and stress-related disorders.
-
Remote monitoring and telehealth integration are enhancing patient accessibility.
-
Regional diversification is creating opportunities for untapped healthcare markets.
Market Growth Drivers
Several strong factors are accelerating the Auricular Bioelectronic Devices Market:
-
Technological Integration: AI, IoT, and digital health platforms make devices more adaptable and effective.
-
Growing Disease Burden: Increasing prevalence of neurological and stress-related conditions drives adoption.
-
Shift Toward Preventive Healthcare: Rising awareness of wellness encourages use of bioelectronic devices.
-
Non-Invasive Preference: Patients prefer treatments with fewer risks and no surgical requirements.
-
Supportive Ecosystem: Government initiatives, clinical trials, and private investments strengthen industry growth.
Restraints
Despite the promising outlook, the Auricular Bioelectronic Devices Market faces challenges. High development costs and strict regulatory requirements slow down innovation. Lack of awareness in emerging economies reduces adoption, while competition from pharmaceuticals remains strong due to their established market dominance.
Technical issues also present obstacles. Devices must overcome limitations in battery performance, long-term comfort, and user compliance. Furthermore, skepticism among some clinicians about bioelectronic therapies continues to limit widespread acceptance.
Opportunities
The Auricular Bioelectronic Devices Market holds vast opportunities for growth. Personalized bioelectronic therapies represent one of the most promising avenues, as customization improves treatment outcomes and patient satisfaction. The wellness and consumer health segment also provides an untapped market, with auricular devices being positioned as preventive tools for stress relief and relaxation.
Emerging markets such as Asia-Pacific and Latin America present new growth horizons due to increasing healthcare spending, rising awareness, and large patient populations. Collaborations between medical technology companies and AI developers can further accelerate innovation and expand clinical applications.
Regional Insights
The Auricular Bioelectronic Devices Market shows diverse regional dynamics:
-
North America: Currently dominates due to advanced healthcare systems, strong R&D investments, and rapid adoption of medical innovations.
-
Europe: Benefits from a favorable regulatory environment and growing clinical research into bioelectronic medicine. Adoption is increasing in countries like Germany, France, and the U.K.
-
Asia-Pacific: Expected to be the fastest-growing region, driven by rising healthcare expenditure, growing middle-class populations, and government investments in medical technologies. China and Japan are key growth drivers.
-
Latin America and Middle East & Africa: Still emerging but showing strong potential as awareness increases and healthcare infrastructure improves.
Get Free Sample @ https://www.precedenceresearch.com/sample/6885
Auricular Bioelectronic Devices Market Companies
- NeuroSky, Inc.
- Cala Health, Inc.
- Bioness Inc.
- Nurotron Bioelectronics
- Medtronic plc
- Abbott Laboratories
- Cerbomed GmbH
- Biovotion AG
- Boston Scientific Corporation
- Cyberonics (LivaNova)
- SetPoint Medical
- Neuros Medical, Inc.
- Inspire Medical Systems
- Otivio
- ElectroCore, Inc.
- Axonics Modulation Technologies
- Valtronic SA
- Earlogic Devices
- Somavert Bioelectronics
- NeuroSigma, Inc.
Recent Developments
- In July 2025, SetPoint Medical announced the FDA approval of its implantable vagus nerve stimulation implant in the treatment of inflammatory diseases such as rheumatoid arthritis. The approval also facilitated the options of less invasive ones, such as the solutions of neuromodulation based on the ear.
- In September 2024, Apple was cleared to use the AirPods Pro 2 software in an upgraded form that includes health monitoring features of medical-grade quality by the FDA. The ruling established a regulatory and consumer precedent of how healthcare technology will be incorporated into daily personal audio devices
Also Read@ https://www.dailytechbulletin.com/semiconductor-wafer-transfer-robots-market/
