Clinical Trial Materials Manufacturing Market Overview
The clinical trial materials manufacturing market plays a foundational role in the success of pharmaceutical research. From early-phase trials to late-stage drug testing, the manufacturing and distribution of trial materials ensure that compounds are available in the right quantity, formulation, and timeline to support efficient study execution. This market includes the production of investigational drugs, placebo materials, packaging, labeling, and cold chain logistics—all tailored to rigorous regulatory requirements and tight timelines.
The scope of the clinical trial materials manufacturing market has expanded significantly as pharmaceutical companies pursue more complex trials across multiple geographies. Innovations in personalized medicine, adaptive trials, and rare disease research have increased the demand for customized, flexible, and highly compliant manufacturing services.
AI and Innovation in Clinical Trial Materials Manufacturing
Artificial intelligence (AI) is transforming the clinical trial materials manufacturing market by streamlining forecasting, inventory management, and quality control. AI-powered platforms can predict demand patterns based on trial protocols, helping manufacturers avoid delays due to material shortages or overproduction.
Innovations in digital twins, simulation software, and machine learning algorithms are optimizing supply chain processes, allowing trial sponsors to model different manufacturing and logistics scenarios. Moreover, smart packaging and digital labeling powered by blockchain enhance tracking and traceability, ensuring compliance in real time.
Future Trends in the Clinical Trial Materials Manufacturing Market
As clinical trials become more global and personalized, the clinical trial materials manufacturing market is adapting to several emerging trends. One major shift is the move toward decentralized clinical trials (DCTs), which require flexible packaging and direct-to-patient logistics.
Additionally, demand for just-in-time (JIT) manufacturing is increasing to accommodate smaller patient populations and faster trial timelines. Modular manufacturing facilities and micro-batching capabilities are gaining popularity to meet these needs. Sustainability is also becoming a priority, prompting the development of eco-friendly packaging and leaner supply chains across the clinical trial materials manufacturing market.
Rising Demand for Clinical Trial Materials
The demand within the clinical trial materials manufacturing market is climbing steadily due to the increase in clinical trial activity across therapeutic areas such as oncology, immunology, neurology, and infectious diseases. As pharmaceutical companies expand their R&D pipelines, the volume and complexity of clinical trial materials also grow.
Biologics and cell and gene therapies require specialized storage and manufacturing conditions, further fueling demand for sophisticated material preparation and handling. Additionally, regulatory tightening around labeling, serialization, and GMP compliance is pushing sponsors to collaborate with specialized manufacturing partners.
Key Market Highlights
Several important developments are reshaping the clinical trial materials manufacturing market. Leading contract manufacturing organizations (CMOs) are investing in advanced cleanroom facilities, real-time data analytics, and automated packaging lines to improve turnaround and reduce human error.
Innovations in temperature-sensitive shipping solutions and global cold chain logistics have helped sponsors manage complex trials in diverse locations. Companies are also prioritizing sustainability by using recyclable materials, reducing packaging waste, and minimizing emissions associated with transport.
Market Growth Drivers
Key factors driving the clinical trial materials manufacturing market include the rapid expansion of the global pharmaceutical pipeline, increasing complexity in clinical trial protocols, and growing outsourcing trends. Sponsors are increasingly relying on external manufacturing partners to streamline operations and meet accelerated trial timelines.
The proliferation of orphan drug designations and precision medicine also contributes to rising demand. These therapies often require small, highly tailored material batches, pushing the need for agile and responsive manufacturing systems. Regulatory harmonization across regions is another enabler of growth, allowing smoother multinational trial execution.
Restraints in the Market
Despite its upward trajectory, the clinical trial materials manufacturing market faces several restraints. Supply chain disruptions—especially post-pandemic—remain a concern, affecting raw material availability and logistics reliability.
Strict regulatory environments across different jurisdictions complicate manufacturing consistency, while variability in labeling requirements leads to added costs and delays. Capacity constraints among niche manufacturers and limited availability of GMP-certified facilities can also pose challenges to smaller biotechs and emerging sponsors.
Opportunities in the Market
There are many opportunities in the clinical trial materials manufacturing market, particularly in the expansion of services for small and mid-sized biopharma companies. By offering end-to-end solutions, manufacturers can cater to sponsors lacking in-house expertise.
Emerging markets in Asia-Pacific and Latin America are witnessing increased clinical trial activity, opening new demand centers for localized manufacturing hubs. Additionally, technology integration such as AI-driven quality control, smart warehousing, and predictive inventory systems offers a competitive edge for forward-thinking players in this space.
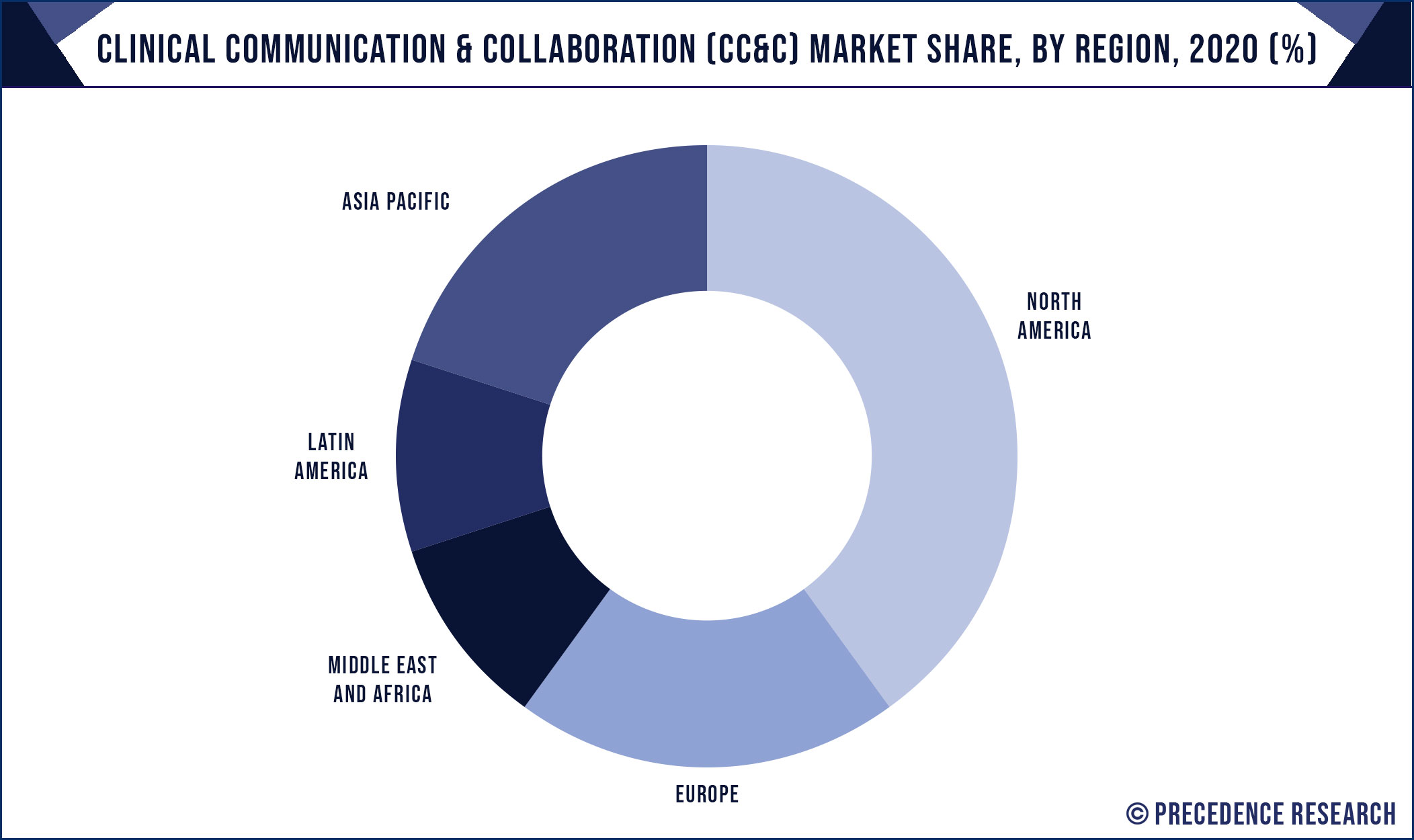
Regional Insights
North America continues to lead the clinical trial materials manufacturing market due to high R&D investments, regulatory clarity, and a strong network of contract manufacturers. The U.S. and Canada serve as hubs for innovation and offer robust infrastructure to support large-scale and niche trials alike.
Europe follows closely, with countries like Germany, Switzerland, and the UK hosting GMP-compliant facilities and prominent CMOs. The Asia-Pacific region is experiencing rapid growth, especially in China, India, South Korea, and Singapore, where lower costs and government support make the region attractive for outsourcing.
Latin America and Africa are emerging as new frontiers, with growing investments in clinical research infrastructure and training. These regions offer cost advantages and diverse patient populations, presenting strong potential for localized material production and logistics.
Also Read@ https://www.dailytechbulletin.com/semiconductor-wafer-transfer-robots-market/
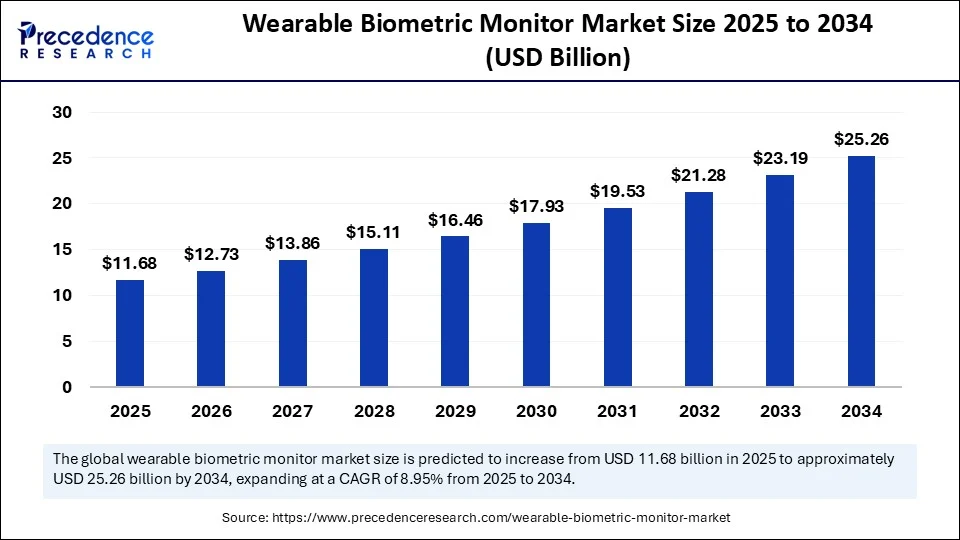
Wearable Biometric Monitor Market Companies

- Abbott (FreeStyle Libre wearable tech)
- ActiGraph
- Apple Inc.
- BioIntelliSense
- Biostrap
- Cardiac Insight
- Empatica
- Fitbit (Google)
- Garmin Ltd.
- Huawei Technologies
- Oura Health (Oura Ring)
- Philips Healthcare
- Polar Electro
- Samsung Electronics
- Valencell
- VitalConnect
- Whoop Inc.
- Withings
- Xiaomi Corporation
- Zephyr Technology (Medtronic)
Get Free Sample Link @ https://www.precedenceresearch.com/sample/6684