Market Overview
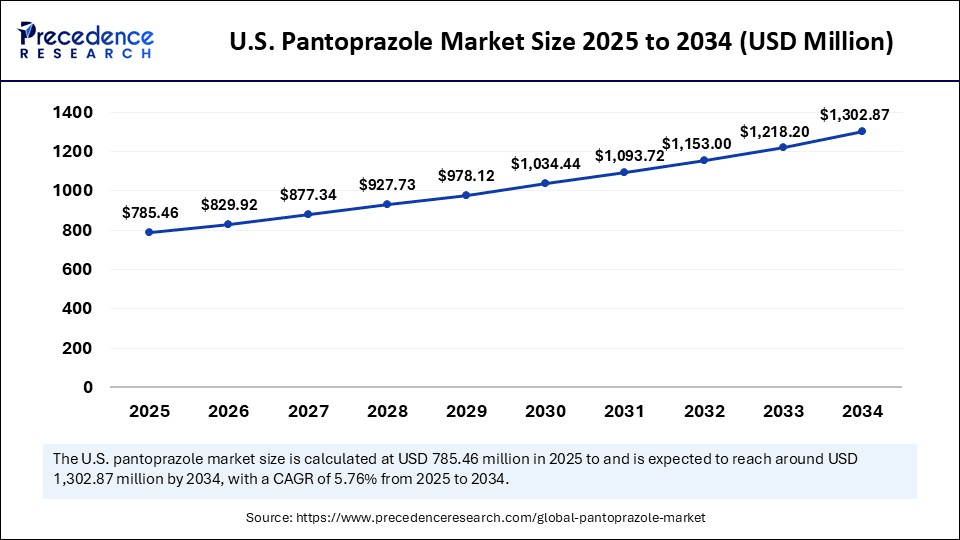
The U.S. Pantoprazole Market is witnessing steady growth due to increasing prevalence of acid reflux and related gastrointestinal disorders. In the Pantoprazole Market in the United States, both branded and generic drugs play a significant role. Patient awareness and prescription patterns are key factors shaping this evolving market landscape.
AI and Innovation
Artificial intelligence is transforming the U.S. Pantoprazole Market by optimizing drug development and supply chain management. Within the Pantoprazole Market in the United States, AI-driven analytics predict demand trends with greater accuracy. Innovation in drug formulation and delivery is accelerating growth and improving patient outcomes.
Future Trends of Market
Future trends in the U.S. Pantoprazole Market include expanding generic drug penetration and rising over-the-counter sales. The Pantoprazole Market in the United States is also expected to benefit from telemedicine’s growing role in prescriptions. Sustainability initiatives and personalized medicine will further influence market dynamics.
Rising Demands of Market
The demand for Pantoprazole is rising sharply in the U.S. Pantoprazole Market due to lifestyle changes, aging population, and increased gastrointestinal conditions. The Pantoprazole Market in the United States sees a surge in both preventive and therapeutic use. These factors contribute to the expanding footprint of Pantoprazole products.
Key Market Highlights
Key highlights of the U.S. Pantoprazole Market include regulatory approvals, new product launches, and strategic partnerships. In the Pantoprazole Market in the United States, competition between branded and generic manufacturers drives innovation. Market players are also focusing on enhancing patient accessibility and affordability.
Market Growth Drivers
Growth drivers in the U.S. Pantoprazole Market are predominantly demographic shifts, healthcare expenditure, and rising chronic disease prevalence. The Pantoprazole Market in the United States also benefits from improved insurance coverage and digital health platforms. These elements collectively stimulate demand and expand market reach.
Restraints
Several restraints impact the U.S. Pantoprazole Market, including safety concerns regarding long-term use and regulatory challenges. The Pantoprazole Market in the United States faces competition from alternative acid-reducing therapies. Price erosion due to generic competition also poses challenges for market growth.
Opportunities
The U.S. Pantoprazole Market presents opportunities through increasing e-commerce sales and telehealth services. The Pantoprazole Market in the United States could expand by targeting underserved demographics and personalized treatments. Advancements in AI offer further prospects for market innovation and efficiency.
Regional Insights
Regional insights reveal that states like California and Texas dominate the U.S. Pantoprazole Market due to population density and healthcare infrastructure. The Pantoprazole Market in the United States shows varying growth rates across urban and rural areas. Local policies and healthcare initiatives significantly influence regional demand patterns.
U.S. Pantoprazole Market Companies
- Pfizer Inc.
- Dr. Reddy’s Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Teva Pharmaceutical Industries Ltd.
- Aurobindo Pharma Ltd.
- Mylan N.V. (Viatris)
- Sandoz (Novartis)
- Cipla Ltd.
- Zydus Cadila
- Lupin Limited
- Torrent Pharmaceuticals
- Siegfried Holding AG
- Glenmark Pharmaceuticals
- Hetero Drugs Ltd.
- Alembic Pharmaceuticals Ltd.
- Ipca Laboratories Ltd.
- Bionpharma Inc.
- Kremers Urban Pharmaceuticals
- Accord Healthcare
- Fresenius Kabi
Recent Developments
- On 9 April 2024, Camber Pharmaceutical launched pantoprazole sodium for delayed-release oral suspension, expanding its portfolio with a formulation indicated for GERD, erosive esophagitis, and Zollinger-Ellison Syndrome.
- On 1 April 205, Alembic Pharmaceuticals received final U.S. FDA approval for pantoprazole sodium for injection, 40 mg/vial.
Get Sample Link @ https://www.precedenceresearch.com/sample/6591
