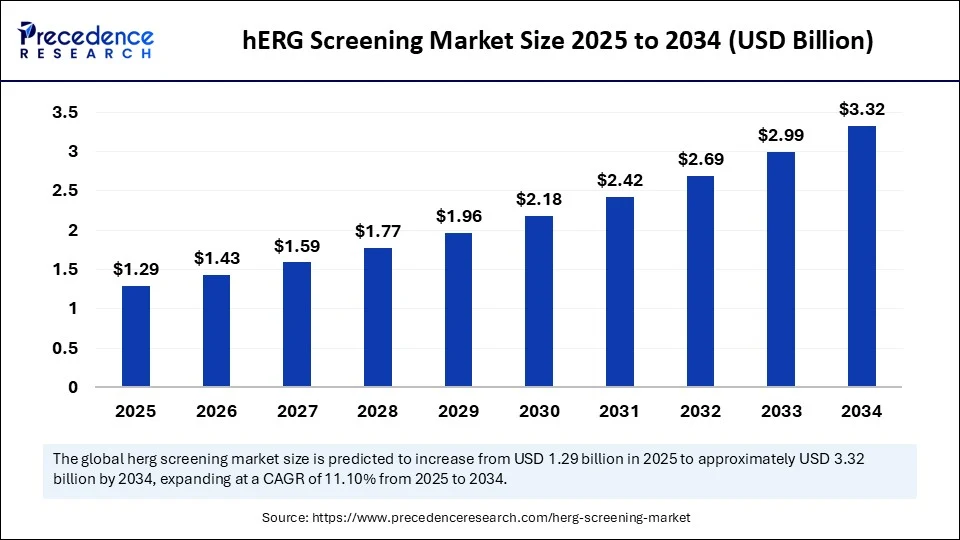
Market Overview
The hERG Screening Market plays a pivotal role in the pharmaceutical and biotechnology industries by helping assess the cardiac safety of new drug candidates. The human Ether-à-go-go-Related Gene (hERG) encodes a potassium ion channel critical for cardiac repolarization. Blockage of this channel can lead to QT interval prolongation, a potentially fatal heart rhythm disturbance.
Due to stringent regulatory frameworks, drug developers are required to conduct thorough screening of compounds for hERG channel interaction. As a result, the hERG Screening Market has become integral to drug discovery, preclinical studies, and regulatory submissions.
Driven by advancements in high-throughput technologies, in vitro assays, and patch-clamp systems, the hERG Screening Market is witnessing rapid evolution. This market is supported by a growing number of pharmaceutical pipelines and an increasing focus on early-stage toxicity screening.
AI and Innovation in the hERG Screening Market
Artificial Intelligence (AI) and machine learning are redefining workflows in the hERG Screening Market. With AI-powered algorithms, it is now possible to predict hERG channel blockade with high accuracy using chemical structure data and known biological responses.
These tools enhance drug safety profiling and reduce false positives, which streamlines the drug discovery process. Furthermore, AI-driven predictive modeling is minimizing the need for extensive in vivo trials, accelerating timelines, and reducing costs in the hERG Screening Market.
Innovation is also evident in automated electrophysiology platforms. Automated patch-clamp systems integrated with AI enable rapid data acquisition and interpretation, drastically improving throughput. Such technological advancements are strengthening the capabilities of the hERG Screening Market and improving regulatory compliance.
Also Read@ https://www.dailytechbulletin.com/herg-screening-market/
Future Trends in the hERG Screening Market
The hERG Screening Market is poised for transformative changes. One key trend is the development of integrated multi-ion channel screening platforms. These next-gen tools not only screen for hERG but also for other cardiac ion channels, providing a holistic cardiac safety profile.
Another emerging trend is the shift toward human-induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs). These cells mimic human cardiac physiology more closely and are gaining traction in the hERG Screening Market for their predictive accuracy and ethical advantage over animal models.
Digital twins and virtual drug screening environments are also anticipated to impact the hERG Screening Market in the near future. These innovations will allow researchers to simulate compound interactions and cardiac effects with minimal wet-lab experimentation.
Rising Demand in the hERG Screening Market
The hERG Screening Market is experiencing surging demand driven by the expansion of drug discovery programs and the growing emphasis on early toxicity testing. As pharmaceutical companies race to develop safer therapeutics, demand for preclinical cardiotoxicity assays continues to climb.
Additionally, the increasing number of regulatory approvals requiring cardiac safety data has made hERG testing indispensable. The hERG Screening Market is also benefiting from rising investments in personalized medicine and oncology drugs, which often carry higher safety risks and require comprehensive preclinical testing.
Contract research organizations (CROs) and academic research institutions are also contributing to this demand, creating new opportunities for vendors in the hERG Screening Market.
Key Market Highlights
The hERG Screening Market is marked by strategic innovations and partnerships. Many leading players are enhancing their assay technologies, launching new high-sensitivity kits, and expanding their service portfolios.
Strategic acquisitions and collaborations between CROs, pharma companies, and technology developers are reshaping the competitive landscape. The increasing adoption of in vitro assays and automated platforms is also a key highlight of the hERG Screening Market.
Moreover, efforts to standardize hERG screening protocols across geographies are ensuring better data comparability and regulatory approval rates. These highlights underscore a robust, rapidly maturing hERG Screening Market.
Market Growth Drivers
-
Regulatory Pressure: Agencies mandate cardiac safety testing for new drug applications, increasing the reliance on hERG assays.
-
Drug Pipeline Expansion: An increase in clinical trials fuels demand for early-stage screening.
-
Technological Advancements: High-throughput screening, AI, and automation drive scalability.
-
Cost Efficiency: Early toxicity detection reduces late-stage failures and development costs.
-
Personalized Medicine: Tailored drugs require deeper safety profiling, boosting the need for cardiac-specific screens in the hERG Screening Market.
Restraints
Despite its expansion, the hERG Screening Market faces certain restraints. One of the key challenges is the high cost of advanced screening equipment, which can be prohibitive for small and mid-sized organizations.
Additionally, the complexity of interpreting hERG assay results can lead to inconsistent data if not standardized properly. False positives may unnecessarily halt the development of promising compounds, affecting overall confidence in the screening process.
Another challenge is the limited availability of skilled professionals capable of managing complex electrophysiological systems, which may slow adoption in some regions of the hERG Screening Market.
Get Sample Link@ https://www.precedenceresearch.com/sample/6531
Opportunities
The hERG Screening Market is ripe with opportunities. The shift toward integrated toxicity platforms that assess multiple cardiac risk factors creates new revenue streams for assay developers and service providers.
Emerging markets present significant untapped potential, particularly in Asia-Pacific and Latin America, where pharmaceutical research is expanding rapidly. There’s also growing opportunity in cloud-based hERG screening data platforms, allowing for remote data interpretation and global collaboration.
Moreover, the rise of non-traditional drug formats, such as biologics and RNA-based therapies, opens new frontiers for hERG-specific risk assessments, broadening the scope of the market
Regional Insights
-
North America: Dominates the hERG Screening Market due to advanced drug development infrastructure, strong regulatory oversight, and widespread use of automation in screening processes.
-
Europe: Focused on ethical screening practices, Europe is increasingly adopting hiPSC models and integrated toxicity platforms, expanding its share in the hERG Screening Market.
-
Asia-Pacific: Rapid growth due to outsourcing trends, rising government support for R&D, and an increasing presence of biotech startups investing in cardiotoxicity screening.
-
Latin America and Middle East & Africa: While smaller in market share, these regions are gaining momentum due to improving healthcare infrastructure and regulatory reforms, presenting long-term opportunities for the market.
hERG Screening Market Companies
- Charles River Laboratories
- Eurofins Scientific
- Cyprotex (Evotec SE)
- Thermo Fisher Scientific
- Metrion Biosciences
- Aurora Biomed
- QPatch (Sophion Bioscience)
- Nanion Technologies
- Axol Bioscience
- B’SYS GmbH
- Multispan Inc.
- ChanTest (now part of Charles River)
- Bioneer Corporation
- Cellectricon
For inquiries about discounts, bulk purchases, or customization options, please reach out to us at sales@precedenceresearch.com
