Blood Transfusion Diagnostics Market Key Takeaways
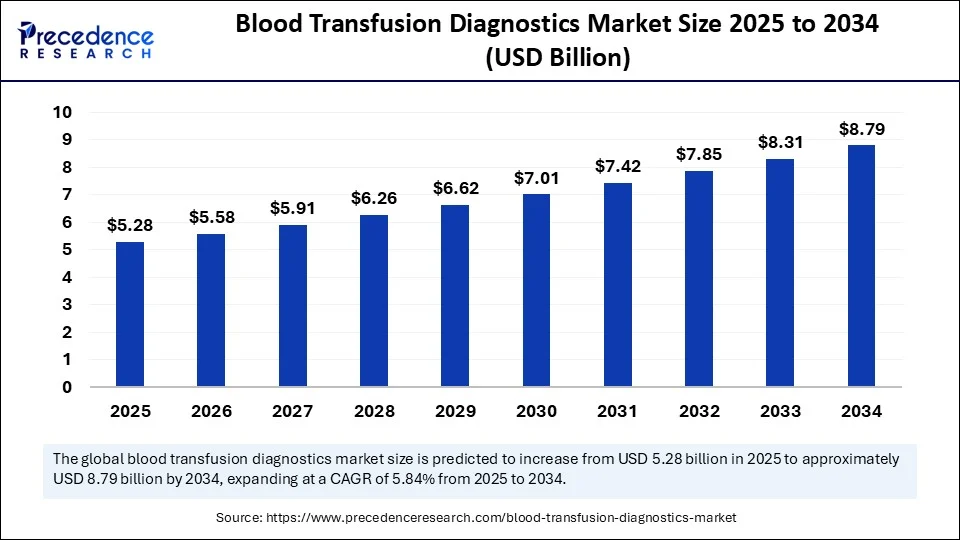
- In terms of revenue, the global blood transfusion diagnostics market was valued at USD 4.98 billion in 2024.
- It is projected to reach USD 8.79 billion by 2034.
- The market is expected to grow at a CAGR of 5.84% from 2025 to 2034.
- North America dominated the blood transfusion diagnostics market with the largest market share of 33.7% share in 2024.
- Asia Pacific is expected to expand at the fastest CAGR between 2025 and 2034.
- By product type, the reagents & kits segment held the biggest market share of 52.8% in 2024.
- By product type, the instruments segment is expected to grow at the fastest CAGR between 2025 and 2034.
- By test type, the disease screening segment captured the biggest market share of 45.1% in 2024.
- By test type, the molecular testing segment is expected to expand at a notable CAGR over the projected period.
- By technology, the serology-based testing segment contributed the major market share of 48.6% in 2024.
- By technology, the molecular-based testing segment is expected to expand at the highest CAGR over the projected period.
- By end user, the hospitals segment generated the major market share of 42% in 2024.
- By end user, the plasma fractionation centers segment is expected to expand at a notable CAGR over the projected period.
How is AI Redefining Blood Transfusion Diagnostics
Artificial intelligence is reshaping the Blood Transfusion Diagnostics Market via smart diagnostics and predictive analytics. AI-enabled modules assist in interpreting complex nucleic acid amplification and serological data, reducing human error and improving test turnaround. Emerging reagent‑free imaging analyzers powered by deep learning offer potential for rapid leukocyte classification without extensive sample preparation. AI is also enabling point-of-care compatibility testing and predictive compatibility scoring, contributing to safer transfusion outcomes and streamlined workflows within diagnostic labs.
Drivers
Key factors fueling the Blood Transfusion Diagnostics Market growth include:
-
Rising prevalence of chronic blood disorders—such as anemia, leukemia, hemophilia—and transfusion-transmitted infections.
-
Growth in surgical and trauma procedures, increasing demand for safe blood compatibility diagnostics.
-
Strong regulatory mandates globally—such as required nucleic acid testing protocols—to mitigate transfusion risks.
-
Expansion of blood banks, transfusion centers, and healthcare infrastructure in emerging economies.
-
Technological advances in automation, molecular testing, and digital integration enhancing reliability and throughput.
Market Trends
Notable trends shaping the Blood Transfusion Diagnostics Market:
-
Shift toward automated blood grouping platforms and computerized cross-matching systems to accelerate workflows.
-
Increased adoption of nucleic acid amplification testing (NAT) for greater sensitivity in infectious-agent detection.
-
Consumables-heavy growth: reagents & kits maintain high share due to repeat usage and lower cost per test.
-
Point-of-care diagnostics popularity is rising, enabling rapid testing during emergencies or at donor centers.
-
Integration of digital information systems (LIS) and automation in high-volume screening laboratories.
Opportunities
Opportunities within the Blood Transfusion Diagnostics Market abound:
-
Growth in emerging markets with expanding healthcare infrastructure and blood safety awareness.
-
Deployment of AI-powered and cloud-connected testing platforms enabling smarter, faster diagnostics.
-
Development of point-of-care microfluidic or portable devices for decentralized blood compatibility testing.
-
Adoption of next-generation sequencing and pathogen inactivation profiling to enhance safety.
-
Collaborative R&D between diagnostic firms and clinical institutions to accelerate validation of advanced assays.
Challenges
Key obstacles facing the Blood Transfusion Diagnostics Market include:
-
High costs of advanced instruments and diagnostic reagents, limiting accessibility in resource‑constrained settings.
-
Shortage of skilled personnel to operate automated platforms and interpret complex molecular tests.
-
Regulatory variation across regions—compliance costs and approval delays hinder adoption.
-
Supply chain disruptions and cost pressure for reagents and kits in global operations.
-
Persistent skepticism over novel test claims without broad clinical validation.
Also Read@ https://www.dailytechbulletin.com/artificial-intelligence-and-analytics-in-surgery-market/
Recent Developments
Recent developments in the Blood Transfusion Diagnostics Market include:
-
In early 2025, a major diagnostics player launched a next-gen automated system offering enhanced speed and accuracy for blood screening.
-
In 2024, leading manufacturers introduced sophisticated digital PCR assays and soluble protein blockers to improve compatibility testing.
-
Late 2023 saw the launch of high‑throughput automated immunohematology analyzers with improved data security and responsiveness.
-
Partnerships between major diagnostic companies enabled distribution of unified QC and data management tools globally.
-
Adoption of AI‑enhanced imaging analyzers and microfluidic test platforms expanded into pilot deployments across labs.
Blood Transfusion Diagnostics Market Companies

- Grifols, S.A.
- Ortho Clinical Diagnostics
- Bio-Rad Laboratories, Inc.
- F. Hoffmann-La Roche Ltd.
- Abbott Laboratories
- Siemens Healthineers AG
- Immucor, Inc.
- Thermo Fisher Scientific Inc.
- Quotient Limited
- Danaher Corporation (Beckman Coulter)
- Becton, Dickinson and Company (BD)
- HemoCue AB
- BioMérieux SA
- DiaSorin S.p.A.
- Sysmex Corporation
- Bag Health Care GmbH
- Fresenius Kabi AG
- Gen-Probe Incorporated (Hologic)
- Agena Bioscience, Inc.
- Tulip Diagnostics (P) Ltd.
Segments Covered in the Report
By Product Type
- Instruments
- Immunohematology Analyzers
- Nucleic Acid Amplification Systems (NAT)
- Microarray Platforms
- ELISA Readers
- PCR Systems
- Others
- Reagents & Kits
- ABO/Rh Typing Reagents
- Antibody Screening Reagents
- NAT Reagents
- HLA Typing Kits
- ELISA Kits
- Others
- Software & Services
- Blood Bank Management Software
- Laboratory Information Systems (LIS)
- Testing Services
- Others
By Test Type
- Blood Group Typing
- ABO Typing
- Rh Typing
- Antibody Screening
- Disease Screening
- HIV
- Hepatitis B & C
- Syphilis
- Malaria
- Other
- Molecular Testing
- NAT (Nucleic Acid Testing)
- Genotyping & HLA Typing
- Pathogen Inactivation Detection
- Crossmatching & Compatibility Testing
- Major Crossmatch
- Minor Crossmatch
By Technology
- Serology-based Testing
- Molecular-based Testing
- Microarray
- Next-generation Sequencing (NGS)
- Enzyme-linked Immunosorbent Assay (ELISA)
- Rapid Diagnostics
- Others
By End User
- Hospitals
- Blood Banks
- Diagnostic Laboratories
- Plasma Fractionation Centers
- Academic & Research Institutes
Others
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Get Sample Link@ https://www.precedenceresearch.com/sample/6520
