Nucleic Acid Aptamers Market Key Takeaways
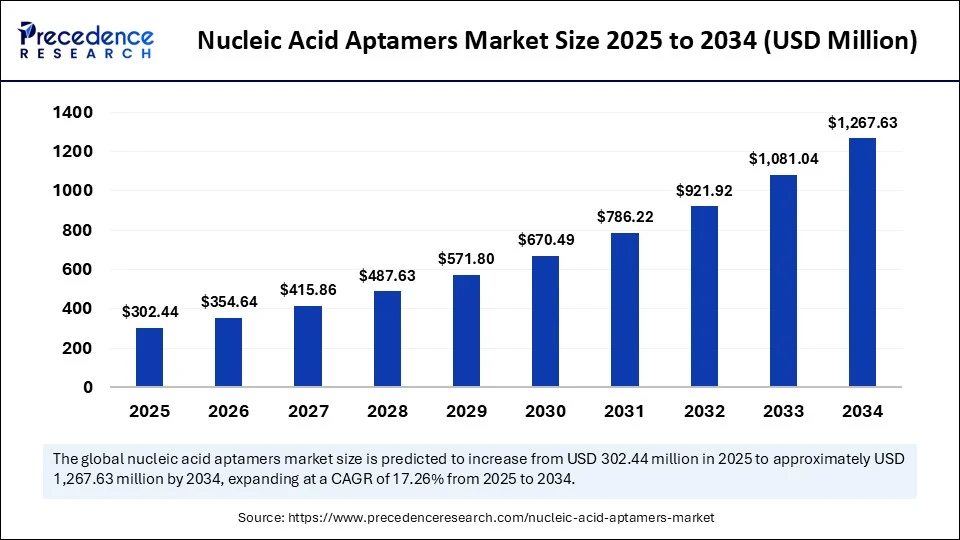
- In terms of revenue, the global nucleic acid aptamers market was valued at USD 257.93 million in 2024.
- It is projected to reach USD 1,267.63 million by 2034.
- The market is expected to grow at a CAGR of 17.26% from 2025 to 2034.
- North America dominated the nucleic acid aptamers market with the largest share of 39% in 2024.
- Asia Pacific is expected to grow at the fastest CAGR from 2025 to 2034.
- By type, the DNA aptamers segment held the highest market share of 45% in 2024.
- By type, the RNA aptamers segment will expand at a significant CAGR between 2025 and 2034.
- By application, the therapeutics segment contributed the biggest market share of 28% in 2024.
- By application, the diagnostics segment will expand at the highest CAGR between 2025 and 2034.
- By synthesis method, the SELEX (systematic evolution of ligands by exponential enrichment) segment led the market, under which the conventional SELEX segment held a significant share as a sub-segment in 2024.
- By synthesis method, the non-SELEX-based methods segment will grow at a significant CAGR between 2025 and 2034.
- By delivery method, the conjugated delivery segment captured the major market share of 41% in 2024.
- By delivery method, the carrier-mediated delivery segment will grow at the highest CAGR between 2025 and 2034.
- By end-user, the pharmaceutical & biotech companies segment generated the major market share of 36% in 2024.
- By end-user, the contract research organizations (CROs) segment will expand at a significant CAGR between 2025 and 2034.
Impact of AI on the Nucleic Acid Aptamers Market
In the Nucleic Acid Aptamers Market, artificial intelligence is revolutionizing how aptamers are discovered, optimized, and deployed. Machine learning and computational modeling now enable rational candidate generation, recognizing structural motifs linked to high binding affinity and specificity.
Techniques rooted in unsupervised learning—such as Potts‑model based sequence generation—allow creation of chemically diverse aptamers that retain functional folds. AI aids in predicting folding structures, off‑target interactions, and stability improvements, vastly reducing the need for iterative lab‑based SELEX rounds. This results in faster, more cost‑effective development of high‑performance nucleic acid aptamers and scaling to diagnostic and therapeutic platforms.
Get Sample link @https://www.precedenceresearch.com/sample/6514
Market Overview
Nucleic Acid Aptamers Market’s key benefits include facile in‑vitro chemical synthesis, structural tunability, minimal immunogenicity, long shelf life, and batch‑to‑batch consistency—crucial for diagnostics and therapeutics.
The market is segmented into diagnostics (biosensors, pathogen detection), therapeutics (targeted drug delivery, controlled release), research & development tools, and others. Nucleic acid aptamers are particularly favored due to their versatility and scalability relative to peptide aptamers or antibodies.
Drivers
1. Rising prevalence of chronic and infectious diseases: Global increases in cancer, cardiovascular, metabolic and infectious conditions fuel demand for precise diagnostics and targeted treatments.
2. Technological improvements in aptamer generation: SELEX and its newer forms (cell‑SELEX, FRELEX, MARAS, particle display) combined with chemical modifications (unnatural bases, stabilizing groups) enhance affinity, stability and target scope.
3. Strong R&D investments and partnerships: Biotechnology, pharmaceutical companies and academia are collaborating aggressively to explore aptamer applications—from diagnostics to therapeutic mosaics.
4. Cost‑effectiveness versus antibodies: Aptamers can be synthesized chemically at scale at lower cost, without cold‑chain logistics, and with consistent quality—making them attractive for scalable healthcare deployment.
Opportunities
-
Diagnostic innovation: Electrochemical aptamer‑based biosensors are enabling real‑time, in‑vivo measurements of critical biomarkers via wearables or lab‑on‑a‑chip systems. These sensors offer rapid response (seconds), high sensitivity, and multiplexing potential.
-
Therapeutics for underserved areas: Therapeutic aptamers targeting ophthalmic conditions (e.g., macular degeneration), cancer, rare diseases, and viral targets represent substantial unmet needs.
-
Integration in drug delivery and tissue engineering: Aptamers can be integrated with hydrogels, nanoparticles, or bioprinted scaffolds for controlled growth‑factor release or targeted delivery in regenerative medicine.
-
AI‑powered aptamer design and screening: Use of predictive modeling and unsupervised sequence generation allows exploration of novel sequence space and accelerated lead identification.
-
Regulatory momentum: With early approvals and clinical trials in sight, regulatory frameworks around aptamer products will mature, easing entry of new products to market.
Challenges
-
Short in‑vivo half‑life and clearance issues: Bare aptamers are rapidly degraded by nucleases and eliminated via kidneys unless chemically modified to resist degradation or avoid filtration.
-
Limited market penetration and awareness: Compared to antibody standards, aptamers are still building clinical credibility and practitioner acceptance.
-
Regulatory uncertainty: Guidelines for aptamer diagnostics and therapies are evolving slowly; developers face complex approval processes.
-
High development and optimization cost: Selection, screening, modification, and scale-up of aptamers involve significant scientific and financial investment.
-
IP and patent complexity: Many core patents cover SELEX methods and sequence libraries; patent challenges and licensing complexities impact commercialization.
-
Competition from established biologics: Monoclonal antibodies, peptides, and protein scaffolds remain entrenched in diagnostics and therapeutic markets and enjoy strong regulatory precedent.
Recent Developments
-
By March 2024, an aptamer company signed a strategic innovation agreement with a major life‑sciences corporation to apply Optimer ligand technology to biosensing platforms.
-
In May 2023, a leading biotech collaboration between gene‑editing and biopharma firms brought aptamer‑based therapeutics into development pipelines for metabolic and rare diseases.
-
A significant acquisition occurred in 2023 when a top aptamer developer acquired another pipeline leader, expanding its diagnostic and therapeutic capabilities.
-
Cutting‑edge research in early 2024 saw the launch of a Xenoligo platform—using unnatural base‑containing aptamers—to target infectious pathogens in partnership with academic institutions.
-
Clinical programs in ophthalmology advanced: a nucleic acid aptamer targeting VEGF for geographic atrophy in age‑related macular degeneration entered Phase 3 under FDA’s Special Protocol Assessment.
-
SELEX innovations such as FRELEX (target‑free partitioning), particle display systems, and chemical extension of nucleotide libraries are enhancing the market’s ability to generate high‑affinity, stable aptamers.
Also read @
Nucleic Acid Aptamers Market Companies

- SomaLogic Inc.
- Aptamer Group PLC
- NOXXON Pharma AG
- Base Pair Biotechnologies Inc.
- Aptamer Sciences Inc.
- AM Biotechnologies LLC
- NeoVentures Biotechnology Inc.
- TriLink BioTechnologies LLC
- Aptus Biotech S.L.
- Aptagen LLC
- Vivonics Inc.
- Aptamer Solutions Ltd.
- AuramerBio Ltd.
- Bio-Techne Corporation
- Novaptech
- IBA GmbH
- ATDBio Ltd.
- Raptamer Discovery Group
- LC Sciences LLC
- Integrated DNA Technologies (IDT)
Segment Covered in the Report
By Type
- DNA Aptamers
- RNA Aptamers
- XNA Aptamers (Synthetic analogs)
- Others (e.g., Spiegelmers)
By Application
- Therapeutics
- Cancer Therapy
- Ocular Diseases
- Cardiovascular Diseases
- Infectious Diseases
- Others
- Diagnostics
- Biomarker Discovery
- Pathogen Detection
- Point-of-Care Testing
- Imaging Agents
- Research & Development
- Target Validation
- Molecular Probes
- Biosensors / Analytical Devices
- Others (e.g., Food Testing, Environmental Monitoring)
By Synthesis Method
- SELEX (Systematic Evolution of Ligands by Exponential Enrichment)
- Conventional SELEX
- Cell-SELEX
- Capillary Electrophoresis-SELEX
- Microfluidic SELEX
- Non-SELEX-Based Methods
By Delivery Method (for therapeutic aptamers)
- Conjugated Delivery (e.g., PEGylated, Nanoparticle-conjugated)
- Unmodified Free Aptamers
- Carrier-mediated Delivery
- Others
By End User
- Pharmaceutical & Biotechnology Companies
- Academic & Research Institutes
- Contract Research Organizations (CROs)
- Diagnostic Laboratories
- Others (Hospitals, Government Labs)
By Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
