Induced Pluripotent Stem Cells Market Key Takeaways
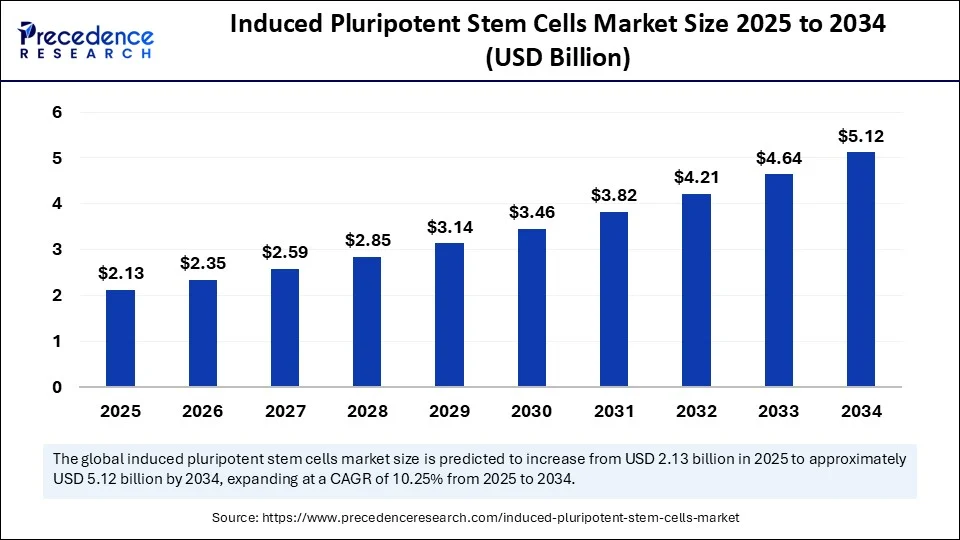
- In terms of revenue, the global induced pluripotent stem cells market was valued at USD 1.93 billion in 2024.
- It is projected to reach USD 5.12 billion by 2034.
- The market is expected to grow at a CAGR of 10.25% from 2025 to 2034.
- North America dominated the global induced pluripotent stem cells market with the largest share of 36% in 2024.
- Asia Pacific is expected to grow at a notable CAGR from 2025 to 2034.
- By application, the drug discovery & toxicology testing segment held the major market share of 36% in 2024.
- By application, the disease modelling segment is projected to grow at a CAGR between 2025 and 2034.
- By cell type/lineage, the hematopoietic cells segment contributed the biggest market share in 2024.
- By cell type/ lineage, the hepatocytes segment is expanding at a significant CAGR between 2025 and 2034.
- By product & services, the iPSC-derived cells segment captured the highest market share of 41% in 2024.
- By product & services, the stem cell banking services segment is expected to grow at a significant CAGR over the projected period.
- By end user, pharmaceutical & biotech companies segments generated the major market share of 44% in 2024.
- By end user, the CROs & CDMOs segment is expected to grow at a notable CAGR from 2025 to 2034.
- By technology, non-integrating methods dominated the global induced pluripotent stem cells market in 2024 and are expected to sustain the growth in the coming years.
Impact of Artificial Intelligence on the Induced Pluripotent Stem Cells Market
Artificial intelligence (AI) is rapidly transforming the induced pluripotent stem cell (iPSC) market by enhancing research efficiency, scalability, and precision in therapeutic development. AI algorithms are instrumental in guiding the reprogramming of adult cells into iPSCs, helping to optimize processes, minimize errors, and improve reproducibility across both academic and commercial settings.
As AI technologies continue to evolve, they open new avenues for scaling clinical applications and accelerating the global adoption of iPSC-based therapies and research tools—bringing regenerative medicine closer to real-world implementation.
Market Overview
The induced pluripotent stem cells market is rapidly expanding as a cornerstone of regenerative medicine, disease modeling, and drug discovery. iPSCs—somatic cells reprogrammed to a pluripotent state—offer ethically-uncontroversial, patient-specific sources for virtually any cell type. Global demand is driven by academic research, pharmaceutical screening, and emerging cell therapy pipelines.
The U.S., Japan, Europe, and China lead in both academic and commercial activity, with research institutes, contract cell‑culture entities, and biotech startups building capacity for iPSC generation, differentiation, and banking services. Forecasts project robust growth throughout the decade, as clinical trials advance and automation scales iPSC-derived products.
Drivers
One powerful driver is therapeutic potential of autologous and allogeneic cell therapies. iPSCs serve as the foundation for personalized regenerative treatments—such as retinal cell replacement, cardiomyocyte delivery, and neuronal regeneration—fueling investment by biopharma. Drug screening and disease modeling also drive growth: iPSC-derived tissue models accelerate small-molecule screening, toxicity testing, and predictive safety, reducing drug development attrition.
Platform improvement and automation: Advances in automated reprogramming workflows, cell‑culture bioreactors, and quality control enable scalable, standardized production. Regulatory clarity and supportive policy: Governments and agencies like the FDA and EMA are issuing guidance frameworks to support clinical translation, enabling fast‑track pathways and supporting GMP manufacturing of iPSC lines.
Opportunities
Regenerative medicine pipelines: As therapeutic applications mature, iPSC-based therapies for macular degeneration, spinal cord injury, and heart failure move toward commercialization. Contract manufacturing services: CMOs specializing in GMP-grade iPSC line generation, expansion, and characterization provide outsourcing opportunities to academia and early-stage biotech. High‑throughput screening platforms: Companies offering differentiated iPSC-derived organoid models (e.g. liver, cardiac, neural) gain traction as pharma seeks predictive human models.
Biobanking and iPSC repositories: Large-scale repositories of disease-specific or ethnically diverse iPSC lines can serve personalized drug screening and translational research needs. AI-powered phenotyping: Integration of image-based machine learning with iPSC-derived tissues enhances throughput, precision, and discovery of novel biomarkers.
Challenges
Despite promise, challenges include manufacturing complexity and standardization. Ensuring iPSC genomic stability, differentiation fidelity, and absence of undifferentiated cells demands stringent quality testing and protocols. High cost and infrastructure demand: GMP-compliant cleanrooms, specialized cell culture media, and skilled technicians impose barriers for new entrants. Regulatory and safety concerns persist: Key issues include tumorigenicity risks, immune rejection in allogeneic settings, and clonal variability.
Ethical/legal issues and consent management: Donor consent, privacy, and commercialization rights over derived lines can present governance headaches. Market fragmentation and immature reimbursement models: No standardized pricing or reimbursement exists for iPSC-based therapies, and clinical adoption depends on demonstration of long-term efficacy and safety.
Recent Developments
In recent years, several biotech firms have entered late-stage clinical trials using iPSC-derived retinal pigment epithelial (RPE) cells to treat macular degeneration and Stargardt’s disease, reporting early promising safety and visual improvement signals.
Multiple contract development and manufacturing organizations (CDMOs) have announced expansion of cGMP iPSC manufacturing suites, offering scale-up from research to clinical-grade product. Big pharma partnerships emerged, marrying iPSC‑based cardiac toxicity platforms with machine‑learning analytics to predict drug-induced arrhythmias.
Standardization consortia have been launched to define quality metrics for iPSC line derivation, differentiation, and genomic screening. Some venture-backed startups introduced automated robotic reprogramming platforms to reduce time and variability in iPSC generation. Academic‑industry joint ventures have produced large-scale iPSC biobanks of disease‑specific lines—for neurological disorders and rare genetic conditions—driving personalized disease modeling and early drug screening.
Induced Pluripotent Stem Cells Market Companies
- Allele Biotechnology & Pharmaceuticals
- Axol Bioscience Ltd.
- bit.bio (synthetic biology iPSC platform)
- BlueRock Therapeutics (Bayer)
- BrainXell, Inc.
- Cellular Dynamics International (Fujifilm Group)
- Century Therapeutics
- Corning Incorporated
- Evotec SE
- Fujifilm Cellular Dynamics, Inc.
- Lifeline Cell Technology
- Lonza Group AG
- Ncardia
- Newcells Biotech
- Pluricell Biotech
- REPROCELL Inc.
- Stemcell Technologies Inc.
- Sumitomo Pharma Co., Ltd.
- Takara Bio Inc.
- Thermo Fisher Scientific, Inc
Latest Announcements by Industry Leaders
Dr. Chikafumi Yokoyama, CEO of REPROCELL Inc.
- In May 2025, REPROCELL is a global leader in providing products and services to support stem cells for clinical and research use. As part of this, REPROCELL launches StemEdit™ Human iPSC non-HLA class 1 (B2M Homo KO) and StemEdit™ Human iPSC non-HLA class 1/2 (B2M/CIITA Homo double KO) cell lines. These cell lines, though intended for research use only, originate from StemRNA Clinical induced pluripotent stem cells (iPSCs) from a healthy donor utilizing our StemEdit™ gene editing technology. “The release of these new hypoimmune StemEdit™ Human iPSC lines represents a key milestone in enhancing immune research and regenerative medicine. By allowing researchers to investigate and modify immune cell interactions with unmatched precision, these cell lines demonstrate REPROCELL’s dedication to supporting scientific progress. We will continue to advance and offer world-class tools to drive innovation in cell therapy and immunology.”
Segments covered in the Report
By Application
- Drug Discovery & Toxicology Testing
- High-throughput drug screening
- Hepatotoxicity and cardiotoxicity testing
- Disease Modeling
- Neurological diseases (ALS, Parkinson’s, Alzheimer’s)
- Cardiovascular and metabolic diseases
- Regenerative Medicine
- Cell-based therapies (retinal, cardiac, hematopoietic)
- Stem Cell Banking
- Academic Research
- Gene Editing Research (iPSC + CRISPR/Cas9)
By Cell Type / Lineage
- Hematopoietic Cells
- Cardiomyocytes
- Neural Stem Cells
- Hepatocytes
- Retinal Pigment Epithelium (RPE)
- Pancreatic Beta Cells
- Chondrocytes & Osteoblasts
- Others (T cells, epithelial cells, etc.)
By Product & Service
- iPSC-Derived Cells
- iPSC Reprogramming Kits & Culture Media
- iPSC Lines & Custom Cell Engineering Services
- CRISPR & Genetic Modification Tools
- Stem Cell Banking Services
- Assay Kits for Differentiation, QC, and Validation
- Instruments & Bioreactors
By End User
- Pharmaceutical & Biotech Companies
- Academic & Research Institutions
- CROs & CDMOs
- Cell Therapy Companies
- Government & Non-Profit Organizations
By Technology
- Integrating Vectors (e.g., Retroviruses)
- Non-Integrating Methods
- Sendai virus
- Episomal vectors
- mRNA & Protein-based reprogramming
- CRISPR-Cas9 & Gene Editing
- Automated iPSC Production Platforms
By Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
Get Free Sample@ https://www.precedenceresearch.com/sample/6503
