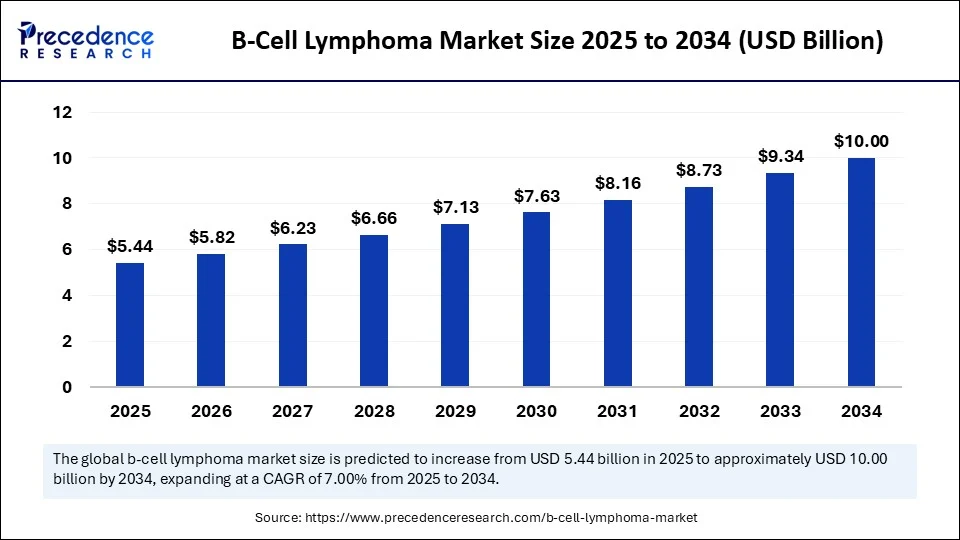
According to Precedence Research, the global B-cell lymphoma market size is estimated at USD 5.44 billion in 2025 and is forecasted to reach around USD 10 billion by 2034, growing at a CAGR of 7.00% from 2025 to 2034. This growth is fueled by advancements in targeted monoclonal antibodies, CAR T-cell therapies, rising awareness, and increasing cancer burden globally.
As healthcare systems focus on personalized medicine and better patient outcomes, the demand for advanced B-cell lymphoma diagnostics and therapies is surging. Key players are investing heavily in immuno-oncology and biomarker research to offer tailored solutions.
Quick Insights: B-cell Lymphoma Market
-
Market Size (2023): USD 21.25 Billion
-
Forecast (2032): USD 33.62 Billion
-
CAGR (2023–2032): 5.13%
-
Dominant Region: North America
-
Fastest Growing Region: Asia Pacific
-
Top Players: F. Hoffmann-La Roche Ltd., Novartis AG, Merck & Co., Bristol-Myers Squibb, Gilead Sciences
-
Top Treatment Types: Monoclonal Antibody Therapy, Chemotherapy
-
Leading End-Use: Hospitals and Clinics
Get a Sample: https://www.precedenceresearch.com/sample/6471
What’s Driving the Market’s Growth?
The rise in B-cell lymphoma incidences, particularly diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma—has prompted significant R&D activities. Immunotherapies, such as CAR T-cell therapies (e.g., Yescarta, Kymriah), and CD20-targeting monoclonal antibodies are revolutionizing the standard of care. Moreover, increased accessibility to advanced diagnostics and growing investments in clinical trials are further propelling market growth.
Aging populations in developed countries, growing healthcare expenditures, and supportive government initiatives are also reinforcing market expansion.
What Trends and Opportunities are Transforming the B-cell Lymphoma Landscape?
-
Precision Oncology at the Forefront: Biomarker-driven treatment selection and genomic profiling are reshaping therapeutic approaches.
-
Rise in CAR T-cell Commercialization: Expanding indications and improved manufacturing scalability for CAR T-cell therapies offer lucrative potential.
-
Combination Therapy Trials: Integration of immunotherapy with standard regimens opens new frontiers.
-
AI in Hemato-Oncology: Emerging tools help in early diagnosis, risk stratification, and treatment personalization.
-
Expansion in Emerging Markets: Government-funded oncology care and rising medical tourism in Asia-Pacific offer untapped potential.
Expert Take: What Industry Leaders Are Saying
“The B-cell lymphoma market is transitioning into an era of tailored, patient-centric care. We are witnessing an inflection point where real-world data, companion diagnostics, and biologic innovation are aligning to unlock remarkable therapeutic gains,”
— Dr. Rhea Deshmukh, Principal Consultant – Oncology Research, Precedence Research
Market Scope
| Report Coverage | Details |
| Market Size by 2034 | USD 10 Billion |
| Market Size in 2025 | USD 5.44 Billion |
| Market Size in 2024 | USD 5.08 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 7.00% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Disease Type, Therapy Type, Route of Administration, End-User Setting, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Regional Analysis: Where Is Growth the Strongest?
North America: Market Leader (Over 38% Share in 2023)
With robust clinical trial pipelines, early adoption of immunotherapies, and favorable reimbursement policies, the U.S. leads the charge. Notably, Medicare coverage for advanced lymphoma therapies enhances access for elderly populations.
Asia Pacific: Fastest Growing Region (CAGR >6.5%)
Rapidly modernizing healthcare systems, increasing cancer awareness, and expansion of multinational pharma operations across China, India, and Japan are catalyzing regional growth. Government policies such as China’s Healthy China 2030 and India’s Ayushman Bharat are improving treatment access.
Europe: Strong Pipeline and Orphan Drug Support
European nations benefit from EMA-backed orphan drug designation and pan-EU regulatory harmonization, promoting B-cell lymphoma innovation.
Segmentation Analysis: A Closer Look at Growth Drivers Across Categories
By Type
The Diffuse Large B-cell Lymphoma (DLBCL) segment commands the largest share of the global B-cell lymphoma market. Accounting for nearly 40% of all B-cell lymphomas, DLBCL is recognized for its aggressive clinical behavior and need for prompt treatment. Innovations such as first-line combination therapies (e.g., Polivy + R-CHP) and CAR T-cell therapies have significantly improved prognosis, further boosting demand in this segment. Additionally, the high prevalence and increasing inclusion of DLBCL in global clinical trials enhance its contribution to market revenues.
Follicular Lymphoma, a more indolent yet recurrent form, holds the second-largest market share. While traditionally treated with chemoimmunotherapy, newer approaches like bispecific antibodies and maintenance therapies are emerging. Increased understanding of its molecular biology is driving more targeted R&D. Mantle Cell Lymphoma (MCL) and other rarer subtypes—such as marginal zone lymphoma—are witnessing a gradual rise in treatment access and diagnostic accuracy, especially in developed countries, opening up new niche opportunities for pharmaceutical companies.
Read Also: Logic Semiconductor Market Set to Cross $231.38 Billion by 2034
By Treatment Type
In terms of treatment modalities, monoclonal antibody therapy leads the market owing to the wide-scale adoption of anti-CD20 antibodies like Rituximab and Obinutuzumab. These therapies have become first-line options due to their efficacy and tolerability, and are often used in combination with chemotherapy. Their dominance is further supported by established reimbursement frameworks and clinical familiarity among oncologists.
Chemotherapy continues to play a central role, particularly in combination regimens for both aggressive and indolent forms. While newer therapies are emerging, chemotherapy remains an essential backbone in many global treatment guidelines, especially in resource-limited settings.
CAR T-cell therapies are the fastest-growing segment. Innovations such as Kymriah and Yescarta have transformed treatment for relapsed/refractory DLBCL and are being expanded to earlier lines and new subtypes. Their personalized nature, high response rates, and growing acceptance in real-world settings are fueling their market share, despite their high cost and manufacturing complexity.
Stem cell transplantation also contributes meaningfully to the market, especially for eligible younger patients, though it faces limitations due to procedural risk and availability. Other treatments—including radioimmunotherapy, kinase inhibitors, and biosimilars—form an important and growing subset of the therapeutic landscape.
By End-Use
When segmented by end-use, hospitals and clinics dominate the market. These institutions handle the majority of B-cell lymphoma diagnoses and treatments, thanks to their access to multidisciplinary oncology teams, infusion centers, and advanced diagnostic infrastructure. Their dominance is further cemented by the ability to conduct and enroll patients in clinical trials, particularly in the U.S. and Europe.
Specialty cancer centers, on the other hand, are gaining traction for their personalized and high-touch care models. With the expansion of standalone cancer hospitals in urban regions and medical tourism hubs—especially in Asia-Pacific—the role of these centers is becoming increasingly pivotal. They often act as centers of excellence for CAR T-cell therapy and advanced biologics administration. The growing demand for outpatient infusion services and patient navigation programs is also elevating their position within the care continuum.
Recent Breakthroughs from Key Market Players
-
Gilead Sciences reported expanded use of Yescarta in second-line DLBCL treatment.
-
Roche’s Polivy, in combination with R-CHP, received FDA accelerated approval as first-line DLBCL therapy.
-
Bristol-Myers Squibb continues CAR T-cell development with Breyanzi for earlier-line use.
-
Novartis AG announced real-world outcomes data showing durable response rates with Kymriah.
-
Merck & Co. is exploring PD-1 inhibitors in combination regimens for relapsed patients.
Challenges and Cost Pressures: What’s Holding the Market Back?
While technological strides are evident, the B-cell lymphoma market faces hurdles such as:
-
High Therapy Costs: CAR T-cell treatments can cost upwards of $400,000 per patient, straining both public and private payers.
-
Complex Manufacturing: Personalized therapies like CAR T-cells require logistical precision and specialized facilities.
-
Regulatory Delays in Emerging Markets: Inconsistent frameworks and limited biologics expertise delay drug access.
-
Post-treatment Relapse Risk: Some patients experience recurrence even after cutting-edge treatments, prompting the need for long-term monitoring solutions.
Case Study: Yescarta’s Transformational Journey in DLBCL
When Gilead’s Yescarta received FDA approval for relapsed/refractory DLBCL, it marked a milestone in personalized immunotherapy. A U.S. cancer center reported over 60% complete remission rate among high-risk patients in its observational study. Yescarta’s success catalyzed a new wave of CAR T-cell research globally, proving its clinical and commercial viability.
