Pharmacovigilance and Drug Safety Software Market Key Highlights
-
North America led the market with a dominant share of 51.50% in 2024, while the Asia Pacific region is projected to grow at the fastest pace over the forecast period.
-
By software type, adverse event reporting software held the largest share of 38.50% in 2024, while signal detection software is expected to witness significant growth during the forecast period.
-
In terms of delivery mode, the on-premise segment accounted for the highest market share of 46.50% in 2024, whereas the cloud-based software segment is forecasted to grow rapidly through 2034.
-
By functionality, case data entry and processing dominated with a market share of 33.50% in 2024, and the regulatory submission and signal management segment is anticipated to expand at a notable CAGR.
-
Among end users, pharmaceutical and biotech companies generated the largest share of 54.50% in 2024, while regulatory agencies and CROs (Contract Research Organizations) are expected to register the fastest growth in the coming years.
What is Driving the Growth of the Pharmacovigilance and Drug Safety Software Market?
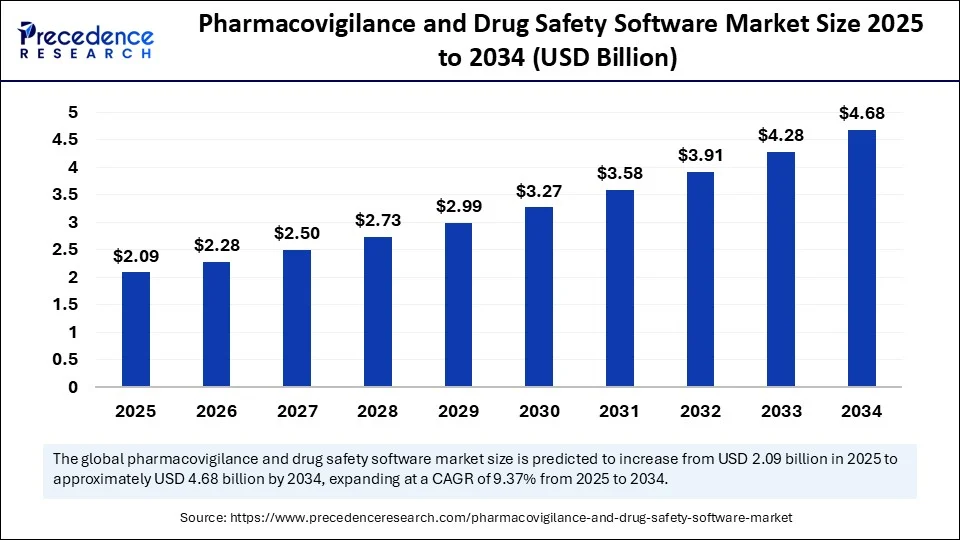
The global pharmacovigilance and drug safety software market is witnessing strong growth, reaching an estimated value of USD 1.91 billion in 2024 and projected to expand to approximately USD 4.68 billion by 2034. With a CAGR of 9.37% expected between 2025 and 2034, the market is gaining momentum as pharmaceutical companies and regulatory bodies increasingly rely on advanced software tools to ensure drug safety and streamline compliance processes.
How is AI Reshaping Pharmacovigilance and Drug Safety?
Artificial intelligence is playing a pivotal role in transforming pharmacovigilance practices by automating complex processes such as case processing, signal detection, and data mining. AI-powered platforms are increasingly being used to analyze data from EHRs, social media, and global safety databases to detect potential adverse drug reactions earlier. This not only accelerates risk mitigation but also enables predictive modeling for better patient outcomes and personalized drug safety assessments.
What Factors Contribute to North America’s Market Dominance?
North America led the global market in 2024, accounting for 51.50% of the revenue share. This leadership is rooted in its advanced pharmaceutical infrastructure, a highly regulated environment, and an early embrace of digital health technologies. The U.S. in particular has been at the forefront, with initiatives like REMS (Risk Evaluation and Mitigation Strategies) from the FDA requiring enhanced drug safety tracking. High investments in R&D and the presence of key global pharma companies further strengthen the region’s leadership.
Why is the U.S. a Strategic Market for Drug Safety Software?
The U.S. remains a key driver of growth in North America due to its large pharmaceutical base and regulatory initiatives. Programs such as OpenFDA and Sentinel demonstrate the government’s commitment to using digital tools for public health monitoring. The launch of new mandates under the REMS program in 2025 is expected to further bolster the adoption of pharmacovigilance software.
What is Fueling Asia Pacific’s Rapid Growth in the Market?
Asia Pacific is poised to grow at the highest CAGR from 2025 to 2034. The region benefits from a surge in clinical trials, growing healthcare investments, and a supportive regulatory environment. Countries such as China, India, and South Korea are emerging as preferred destinations for global drug trials due to cost efficiencies and large patient pools. Additionally, governments are actively promoting digital health solutions and AI-based safety monitoring tools.
What Role Does China Play in Shaping Regional Trends?
China’s vast pharmaceutical landscape and increasing focus on Good Pharmacovigilance Practices (GVP) make it a significant contributor to the regional market. The country is actively reforming its drug safety frameworks and expanding pharmacovigilance to include traditional Chinese medicines, highlighting its efforts to ensure comprehensive drug safety.
Why is Europe a Key Player in Global Pharmacovigilance?
Europe’s emphasis on patient safety, strict pharmacovigilance guidelines, and adoption of technologies such as cloud platforms and real-world evidence (RWE) solutions position it as a crucial region in the market. Countries like Germany, France, and the UK are integrating AI and analytics to enhance real-time safety monitoring across the lifecycle of medications.
Pharmacovigilance and Drug Safety Software Market Scope
| Report Coverage | Details |
| Market Size by 2034 | USD 4.68 Billion |
| Market Size in 2025 | USD 2.09 Billion |
| Market Size in 2024 | USD 1.91 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 9.37% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Software Type, Delivery Mode, Functionality, End User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
What Opportunities Are Emerging in Latin America?
In Latin America, rising awareness of drug safety and modernization in the healthcare sector are boosting the adoption of pharmacovigilance tools. Regulatory agencies in countries like Brazil and Mexico have introduced post-market surveillance mandates, promoting the use of drug safety software. These developments open doors for tech providers to support regulatory and compliance initiatives.
What Is Driving Growth in the Middle East & Africa Region?
Growth in the Middle East & Africa is being supported by expanding pharmaceutical activity, increased clinical trials, and stronger regulatory collaboration. Initiatives such as the Middle East Regulatory Network and efforts by the Saudi Food and Drug Authority (SFDA) are helping the region catch up in terms of regulatory readiness and pharmacovigilance infrastructure.
Ambulatory Infusion Centers Market Size to Exceed USD 126.27 Bn by 2034
Pharmacovigilance and Drug Safety Software Market Trends
What Are the Current Market Trends Influencing Growth?
-
Increased R&D Spending: Rising investments in drug development create more demand for post-market surveillance.
-
Adoption of Cloud-Based Solutions: Cloud software offers scalability, cost-efficiency, and easier compliance management.
-
Growth of Outsourcing Models: CROs and BPOs are investing in PV software to handle increasing safety data volumes.
-
Stricter Regulations: Global authorities are tightening safety requirements, pushing firms to adopt robust systems.
-
AI and ML Integration: These technologies enhance real-time monitoring, pattern recognition, and risk analysis.
What Are the Main Drivers of the Pharmacovigilance and Drug Safety Software Market?
The rising incidence of adverse drug reactions (ADRs) across clinical trials and post-approval phases is the primary driver. Pharmaceutical companies are under pressure to adhere to evolving global safety regulations, prompting widespread software adoption. Moreover, outsourcing safety functions to CROs increases the need for scalable and efficient digital solutions.
What Challenges Could Restrain Pharmacovigilance and Drug Safety Software Market Expansion?
One of the key barriers is the high cost associated with implementing and maintaining pharmacovigilance systems. Smaller firms may struggle with integrating advanced platforms into legacy infrastructure, and ongoing compliance updates often demand significant resource investment. Technical integration with existing healthcare IT systems like EHRs and LIMS also adds complexity.
What Growth Opportunities Are Emerging in the Pharmacovigilance and Drug Safety Software Market?
A major growth opportunity lies in the integration of AI and ML with pharmacovigilance systems. These technologies are helping companies automate ADR detection, streamline regulatory reporting, and reduce operational costs. Furthermore, expansion into emerging markets with growing pharmaceutical pipelines provides a vast, untapped opportunity for software vendors.
Market Segments
Which Software Type Is Leading the Market?
In 2024, adverse event reporting software accounted for 38.50% of the market share, owing to its importance in meeting compliance and managing growing volumes of safety data. These platforms help ensure efficient, error-free reporting to global regulators. Meanwhile, signal detection software is gaining traction for its predictive capabilities and real-time data analytics, making it the fastest-growing segment.
Why Do On-Premise Solutions Continue to Dominate?
The on-premise delivery model held a 46.50% market share in 2024. Organizations favor on-premise setups due to heightened data privacy concerns, control over sensitive information, and tailored integration. Despite this, cloud-based solutions are gaining ground, especially among companies needing scalable and collaborative platforms.
Which Functional Segment Leads the Market?
The case data entry and processing functionality captured 33.50% of the market in 2024. This segment ensures accurate intake and classification of safety data, which is vital for compliance and decision-making. On the other hand, the regulatory submission and signal management segment is anticipated to grow rapidly as real-time monitoring and early intervention become critical.
Who Are the Primary End Users of Pharmacovigilance Software?
Pharmaceutical and biotech companies were the top users, holding 54.50% of the market in 2024. This is driven by a growing drug pipeline and the regulatory need for drug safety monitoring. Meanwhile, CROs and regulatory agencies are emerging as key adopters, fueled by increased outsourcing and a focus on real-time safety analytics.
Pharmacovigilance and Drug Safety Software Market Companies
-
Oracle Health Sciences
-
Veeva Systems
-
IQVIA
-
ArisGlobal
-
AB Cube
-
Sparta Systems (Honeywell)
-
Medidata (Dassault Systèmes)
-
RxLogix
-
Clario (Bioclinica)
-
TCS ADD Safety
-
Cognizant
-
Tech Mahindra (via NVIDIA partnership)
You can place an order or ask any questions, please feel free to contact at sales@precedenceresearch.com |+1 804 441 9344

